A 42-year-old woman was referred to the retina clinic for evaluation of blurred vision and “blackouts” in both eyes. She had a history of migraines since childhood, which were worsening over the past six months. During this time, she experienced decreased hearing, intermittent vertigo lasting minutes and tinnitus. All of her symptoms had been progressively worsening.
An otolaryngologist performed an audiogram and diagnosed her with otosclerosis. An endocrinologist prescribed her amphetamine/dextroamphetamine for her headaches, but this did not help.
Two months earlier, she had a one-hour episode of dysarthria and arm apraxia, whereupon she presented to the emergency department. Brain computed tomography was normal, and she was diagnosed with a complex migraine.
 |
Workup and imaging findings
At her visit in the retina clinic, visual acuity was 20/40 OD and 20/30 OS. Intraocular pressures were normal. The anterior segment was quiet and unremarkable bilaterally. Fundus examination of the right eye disclosed multifocal yellow intra-arterial plaques (Figure 1). The left eye fundus was normal. Widefield fluorescein angiography demonstrated multiple arteriolar occlusions and segmental arteriolar leakage of the right eye and focal non-perfusion of the left eye (Figure 2, page 12). The left eye was normal. Optical coherence tomography of the macula was normal bilaterally.
Hospital admission and labs
She was admitted to the hospital. Brain MRI showed a focal hyperintense signal abnormality on the fluid-attenuated inversion recovery (FLAIR) sequence in the corpus callosum, multiple punctate areas of diffusion restriction in the subcortical white mater, and an old right thalamic infarct (Figure 3, page 12).
Serum laboratory studies were notable for mildly elevated erythrocyte sedimentation rate of 34 mm/hr., but normal C-reactive protein, blood count and electrolyte levels. Also normal were antineutrophil cytoplastic antibodies, antinuclear antibody, anti-double stranded DNA, complement C3 and C4 level and cryoglobulin testing. Lumbar puncture with cerebrospinal fluid analysis revealed normal chemistry, negative encephalitis panel and no oligoclonal bands.
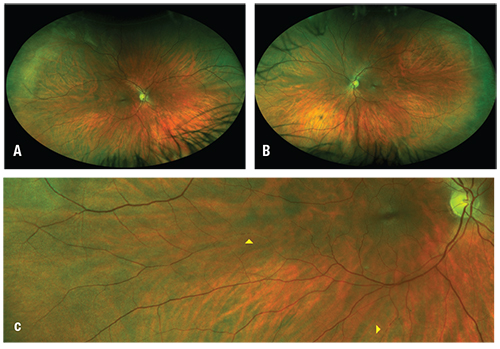 | Figure 1. Widefield fundus photographs of the right (A) and left (B) eyes. Higher magnification (C, inset of A) of the right eye discloses two segments of intra- arterial plaques (arrowheads). |
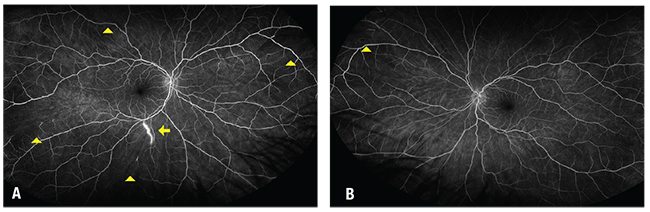 |
| Figure 2. Fluorescein angiogram of the right eye (A) shows arteriolar occlusion in four regions (arrowheads) and arterial wall hyperfluorescence (arrow). The left eye (B) shows focal nonperfusion (arrowhead). |
The diagnosis is ...
Based on the patient’s findings, she was diagnosed with Susac syndrome. Her neurologist agreed that the focal hyperintensity in the corpus callosum was consistent with the diagnosis. She began high-dose oral prednisone 100 mg/day on a tapering regimen managed by her neurologist. She also started taking omeprazole and a calcium/vitamin D supplement.
 |
Follow-up
Her headache and tinnitus resolved. She was seen again two months after her initial presentation at which time she was taking prednisone 60 mg/day. Repeat fluorescein angiography demonstrated new areas of arteriolar occlusion and arterial wall hyperfluorescence (AWH) in the right eye and new AWH in the left (Figure 4).
She also experienced temporary extremity weakness lasting days each time the prednisone dose was decreased. Because of the incomplete response and high dose of prednisone that was required, mycophenolate mofetil 500 mg b.i.d. was started. She tolerated the medication well and has continued tapering the prednisone with plans for serial follow-up and fluorescein angiography.
The Susac constellation
The constellation of hearing loss, encephalopathy, and branch retinal artery occlusions is the classic triad of manifestations in Susac syndrome.1 This case highlights many of the characteristic findings of the disease.
Because Susac syndrome is a rare disease, recognizing it requires a high index of suspicion. The condition is often underdiagnosed, in part because the full triad is found only infrequently when the patient first presents.2 Moreover, BRAOs in these patients may be peripheral and subacute, so patients may not complain of vision loss. Retinal whitening may be absent. Widefield fluorescein angiography has made the detection of these small BRAOs easier.3
In 1979, John Susac, MD, and colleagues reported the first series of this condition in two young women who developed subacute progressive neurologic symptoms and multifocal retinal artery occlusions.4 Based on the ocular findings and brain biopsy of one of the women, which demonstrated angiitis, he concluded the syndrome involved a microangiopathy of the brain and retina.
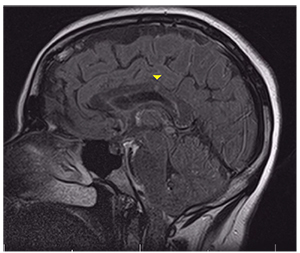 |
| Figure 3. Fluid-attenuated inversion recovery MRI shows a focal hyperintense foci in the corpus callosum (arrowhead). |
Other telltale signs
In 1986, J. Donald Gass, MD, and colleagues described what we now refer to as Gass plaques, which are yellow arteriolar wall deposits often distributed along the mid-segment of the vessel away from the bifurcation points.5,6 Another associated finding is arterial wall hyperfluorescence, a prominent feature in our patient, which can be seen on fluorescein angiography and may not necessarily be located near the BRAOs.7
Brain lesions tend to include small white matter lesions with a special predilection for the corpus callosum. MRI often shows focal hyperintensity on T2 or FLAIR lesions when they are acute and hypointense holes on T1 imaging when they are chronic.8 It is important to distinguish these lesions from those of multiple sclerosis because they can both involve the deep white matter, but demyelination is not typical of Susac syndrome.9
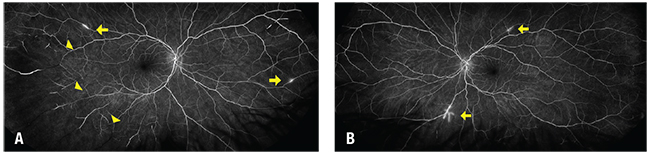 |
| Figure 4. Fluorescein angiogram of the right eye (A) shows increased areas of arteriolar nonperfusion (arrowheads) and new segments of arterial wall hyperfluorescence (AWH, arrow). The left eye (B) also displays new AWH (arrows). |
The exact pathogenesis of Susac syndrome is unclear. It is believed to be an autoimmune process targeting endothelial cells that induce microvascular occlusions of the central nervous system, inner ear and retina. The anti-endothelial response leads to retinal damage ranging from arteriolar plaques to occlusions. Circulating anti-endothelial cell antibodies have been isolated in some patients, but standardized antibody testing has not been developed.10
 |
Treatments
The initial treatment is usually systemic intravenous or oral steroids with the goal to stabilize the ocular and neurologic disease.11 Intravenous immunoglobins have also been used. After treatment of the acute phase, a slow taper of steroid is indicated. Most cases exhibit fairly rapid improvement in headache and neurologic sequelae.
If the disease is persistent or recurrent with steroid tapering, another immunosuppressant such as mycophenolate, azathioprine or cyclophosphamide may be used. Susac syndrome is self-limiting, but it can recur in a minority of patients.
The bottom line
While not all patients have the complete triad on presentation, there are several characteristic findings of Susac syndrome in both the retina and the brain that should prompt evaluation for the disease. It is important for neurologists and otolaryngologists to recognize the disease as well. RS
REFERENCES
1. Vishnevskia-Dai V, Chapman J, Sheinfeld R, et al. Susac syndrome: Clinical characteristics, clinical classification, and long-term prognosis. Medicine (Baltimore). 2016;95:e5223.
2. Dörr J, Krautwald S, Wildemann B, et al. Characteristics of Susac syndrome: a review of all reported cases. Nat Rev Neurol. 2013;9:307-316.
3. Salvanos P, Moe MC, Utheim TP, Bragadóttir R, Kerty E. Ultra-wide-field fundus imaging in the diagnosis and follow-up of Susac syndrome. Retin Cases Brief Rep. 2018;12:234-239.
4. Susac JO, Hardman JM, Selhorst JB. Microangiopathy of the brain and retina. Neurology. 1979;29:313-316.
5. Gass JD, Tiedeman J, Thomas MA. Idiopathic recurrent branch retinal arterial occlusion. Ophthalmology. 1986;93:1148-1157.
6. Egan RA, Hills WL, Susac JO. Gass plaques and fluorescein leakage in Susac syndrome. J Neurol Sci. 2010;299:97-100.
7. Martinet N, Fardeau C, Adam R, et al. Fluorescein and indocyanine green angiographies in Susac syndrome. Retina. 2007;27:1238-1242.
8. Do TH, Fisch C, Evoy F. Susac syndrome: report of four cases and review of the literature. AJNR Am J Neuroradiol. 2004;25:382-388.
9. Buzzard KA, Reddel SW, Yiannikas C, Sean Riminton D, Barnett MH, Hardy TA. Distinguishing Susac’s syndrome from multiple sclerosis. J Neurol. 2015;262:1613-1621.
10. Jarius S, Kleffner I, Dörr JM, et al. Clinical, paraclinical and serological findings in Susac syndrome: an international multicenter study. J Neuroinflammation. 2014;11:46.
11. Grygiel-Górniak B, Puszczewicz M, Czaplicka E. Susac syndrome—clinical insight and strategies of therapy. Eur Rev Med Pharmacol Sci. 2015;19:1729-1735.




