A U-011, Aura Biosciences’ novel viral nanoparticle conjugate candidate, could be the first advance for treatment of ocular cancers since brachytherapy emerged decades ago. The Food and Drug Administration has granted AU-011 orphan drug and fast-track designations.
Conventional treatment for ocular melanoma involves radiotherapy, most commonly in the form of brachytherapy that requires surgical placement of a radioactive plaque on the sclera overlying the choroid. This technique requires multiple operations to place and remove the plaque, and can damage adjacent ocular structures.
AU-011 involves intravitreal injection of the tumor targeted drug, and then uses a laser to activate it. The platform is derived from technology that John Schiller, PhD, of the Center for Cancer Research at the National Cancer Institute had developed.
Preclinical results have demonstrated that AU-011 could selectively destroy ocular melanoma tumors in rabbit eyes.1 At the American Academy of Ophthalmology Retina Subspecialty Day last year, Carol Shields, MD, of Wills Eye Hospital in Philadelphia, reported interim data from the Phase Ib/II trial that showed the drug was well-tolerated at three to six months in the first six patients treated.2 Now, Aura is conducting a dose optimization study with plans to go into Phase III as early as next year.
Here, Aura founder and CEO Elisabet de los Pinos, PhD, and Chief Medical Officer Cadmus Rich, MD, answer questions about AU-011.
The mechanism of action in their own words
Dr. de los Pinos explains AU-011 attaches to heparan sulfate proteoglycans (HSPGs) that are highly expressed and modified on the tumor cell membrane. The viral nanoparticle is just the protein shell that forms the capsid of a virus and it is conjugated to a light activatable cancer drug. It selectively targets and binds the melanoma cells in the eye.
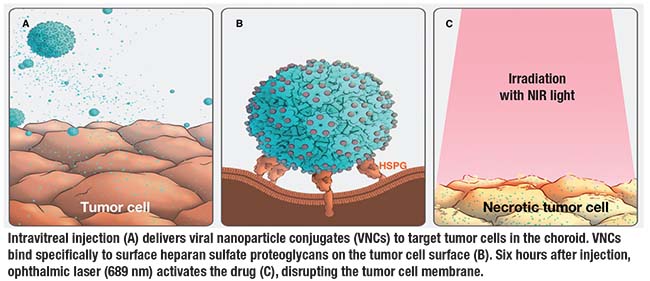 |
Six hours after AU-011 is injected intravitreally, near infrared light is applied with an ophthalmic laser to activate the agent, a process that disrupts the tumor cell membrane. This leads to acute tumor cell necrosis.
The light-activated treatment is double specific: The drug spares surrounding, non-cancerous tissue because it is so specifically targeted to the tumor cells; and the laser light focuses only on the lesion, which ensures the activation of the drug exclusively in the area of the tumor.
Dr. Rich notes that AU-011 has no genetic material, so it will not multiply or spread when injected.
What is the significance of the preclinical data?
Dr. de los Pinos says the goal is to have a highly effective treatment that preserves vision and avoids the comorbidities of radiotherapy. Preclinical studies have reported that AU-011 selectively destroyed choroidal melanoma tumors both in vitro and in vivo in a rabbit model.1 While the limitations of the animal model are well known for demonstrating the efficacy of drugs in other types of cancer, the rabbit model closely replicates the human eye and has more relevance in ocular cancer research. In the rabbit model, the tumor grows in the same layer of the eye—the choroid—as in human pathology.
Highly regarded ocular pathologists from Massachusetts Eye and Ear Infirmary and Emory Eye Center replicated the data in multiple independent studies, Dr. de los Pinos says. Dr. Rich adds that the cells implanted in the rabbit eyes were human melanoma cells, and the preclinical results demonstrated that the drug was able to get to the choroid, bind to human melanoma cells and selectively kill the tumor cells while sparing the adjacent retina.
How does this compare to the current standard of care for ocular melanoma?
Ocular melanoma is the most common primary tumor in the eye with a yearly incidence around 5-6 per million3 and has a mortality rate of 50 percent at 10 years.4 The traditional therapy had been enucleation of the affected eye, Dr. Rich explains.
That changed about 20 years ago when the Collaborative Ocular Melanoma Study (COMS) investigating radiation delivered via iodine125 brachytherapy for choroidal melanoma demonstrated a similar metastatic risk and mortality compared to enucleation.5 The treatment evolved to also include external proton-beam radiation. Both forms of radiotherapy were relatively vision-sparing and eye-sparing compared to enucleation. However, the visual outcomes reported that a majority of patients had significant vision loss.
Since then, the standard of care of these primary tumors has not improved beyond radiotherapy. Different types of radio isotopes have been used, but up to 67 percent of patients will lose vision and 20 percent or more of these individuals will go legally blind in the eye after radiotherapy.6 For large tumors or recurrent melanoma, enucleation is still an option.
What are the risks of radiotherapy for treatment of ocular cancer?
Radiotherapy delivers what Dr. Rich calls “poison” to the eye in the most direct, targeted methods possible—through brachytherapy or external beam therapy. The main problem is that radiation retinopathy, also known as radiation maculopathy, damages the fovea, or radiation papillopathy damages the optic nerve if the radiotherapy is too close to those structures. This can cause severe vision loss.
The operations to place and then remove the brachytherapy plaque, which has radioactive seeds to deliver radiation to the tumors, can require removal of extraocular muscles. That can cause diplopia postoperatively. Cataract and glaucoma are also potential sequelae.
External beam radiation is done through the front of the eye. Known side effects include decreased vision, similar to brachytherapy, along with dry eye, cataracts and neovascular glaucoma. Dr. Rich notes these are not benign treatments, but they do lead to local control of the choroidal melanoma.
How does targeted therapy advance treatment of ocular cancer?
In cancer, the goal is to treat patients early in the disease course. Based on feedback Aura has received from ocular oncologists and retina specialists, AU-011 has the potential to give them a vision-preserving therapy that may be used earlier in the disease course than radiation, which because of its side effects, has typically been used when tumors have documented growth or are a certain size with risk factors. AU-011 has demonstrated a good safety profile that could enable the earlier treatment of smaller ocular tumors while preserving vision for patients, Dr. Rich says.
Where does the ongoing Phase Ib/II trial stand?
Choroidal melanoma is a rare disease, so the trial is using a model with smaller dose-escalation cohorts, Dr. Rich says. The testing involves three ascending single dose treatments followed by light activation with a laser, and results so far show the agent is safe, well tolerated and does not affect vision.
Now the trial is moving into the multiple ascending dose-treatment phase, which is identifying the optimal regimen of AU-011 to maximize the efficacy of the drug while maintaining a good safety profile. Dr. Rich says the results are highly encouraging, but it’s still very early in the trial. RS
REFERENCES
1. Kines RC, Varsavsky I, Choudhary S, et al. An infrared dye-conjugated virus-like particle for the treatment of primary uveal melanoma. Mol Cancer Ther. 2018;17:565-574.
2. Shields CL. A Phase Ib clinical safety study of a novel tumor targeted therapy (AU-011) for the treatment of primary choroidal melanoma. Paper presented at: American Academy of Ophthalmology Retina Subspecialty Day; November 10, 2017; New Orleans, LA..
3. Singh A, Turell M, Topham A. Uveal melanoma: trends in incidence, treatment, and survival. Ophthalmology 2011;118:1881–1885.
4. Rietschel P, Panageas KS, Hanlon C, Patel A, Abramson DH, Chapman PB. Variates of survival in metastatic uveal melanoma. J Clin Oncol. 2005;23:8076–8080.
5. Jampol LM, Moy CS, Murray TG, et al. for the Collaborative Ocular Melanoma Study Group (COMS Group). The COMS randomized trial of iodine125 brachytherapy for choroidal melanoma: IV. Local treatment failure and enucleation in the first 5 years after brachytherapy. COMS report no. 19. Ophthalmology. 2002 ;109:2197-2206..
6. Wisely CE, Hadziahmetovic M, Reem RE, et al. Long-term visual acuity outcomes in patients with uveal melanoma treated with 125I episcleral OSU-Nag plaque brachytherapy. Brachytherapy. 2016;15:12-22.



