| ABOUT THE AUTHORS | |
 | Dr. Jansen (top) and Dr. Tyler (center) are residents in ophthalmology at the University of Texas Health Science Center (UTHSC) San Antonio. Dr. Singer is managing partner of Medical Center Ophthalmology and associate professor at UTHSC San Antonio. DISCLOSURES: Dr. Singer disclosed relationships with Acucela, Alcon, Allergan, Bausch + Lomb, Genentech, Regeneron and Thrombogenics. Drs. Jansen and Tyler had no relationships to disclose. |
 | |
 | |
Combination therapy of anti-vascular endothelial growth factors (anti-VEGF) and sustained-release steroids have emerged as a new approach to treating both branch and central retinal vein occlusions. This article looks at the growing role of combination therapy in the management of these diseases.
Two Arms of Treatment
Treatment of BRVO and CRVO began to change with the introduction of new injectable agents like anti-VEGF and steroids. In 2010, the BRAVO study (for Ranibizumab For The Treatment Of Macular Edema Following Branch Retinal Vein Occlusion) showed that ranibizumab (Lucentis, Genentech) intravitreal injection decreased central foveal thickness (CFT) and, more importantly, restored a mean of 18.2 letters of best-corrected visual acuity (BCVA) at six months with 61.1 percent of patients gaining 15 or more letters at six months.4The CRUISE study (for Ranabizumab For Macular Edema Following Central Retinal Vein Occlusion) also showed a decrease in CFT as well as a mean improvement in BCVA of 14.9 letters at six months, with 47.7 percent of patients gaining 15 or more letters at six months.5
The GENEVA study (for Randomized, Sham-Controlled Trial Of Dexamethasone Intravitreal Implant In Patients With Macular Edema Due To Retinal Vein Occlusion), also showed that sustained-release dexamethasone (Ozurdex, Allergan) was effective in treating macular edema in BRVO and CRVO. Patients showed a mean improvement in BCVA of about 10 letters at 60 days, with 29.3 percent gaining 15 or more letters compared to placebo.6
Role of Steroids in Treatment
The two treatment arms for RVO, steroids and anti-VEGF drugs, work on the associated inflammatory and ischemic processes. Corticosteroids influence multiple inflammatory mediators. The thinking is that steroids may help stabilize tight junctions of retinal vessels and decrease inflammation in the area around occlusions.Steroids suppress many pro-inflammatory mediators, including VEGF, in the eye. In RVO, ischemic cells release growth factors such as erythropoietin and VEGF that increase capillary permeability and cause edema. Anti-VEGF agents decrease swelling and reduce vascular leakage.
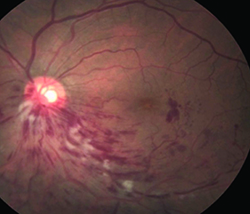 | 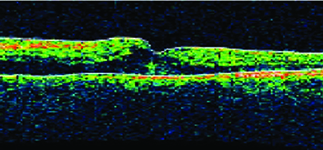 |
| Figure 1. Before combination therapy, retinal imaging shows macular edema in the inferonasal quadrant (left), while optical coherence tomography shows thickening of the central fovea. | |
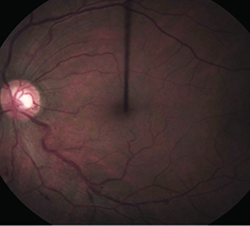 | 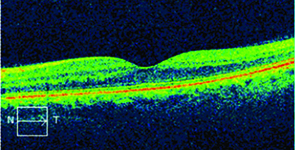 |
| Figure 2. After combination treatment, the macular edema shows significant improvement (left) while optical coherence tomography confirms thinning of the central fovea. | |
Combination Therapy Emerges
An emerging possibility for the treatment of RVO is giving both intravitreal sustained-release dexamethasone and an anti-VEGF agent together, which has been shown to improve visual acuity and prolong the time between injections (reinjection interval).7 Many patients receive these medications together in one way or another when they switched from an anti-VEGF agent to the dexamethasone implant, or vice versa, when they stop responding to the first therapy.A recent subgroup analysis of two or more dexamethasone intravitreal implants given as monotherapy or in combination with an anti-VEGF for treatment of RVO looked at the mean time between injections, as well as the change from baseline for BCVA and CFT.8 The results showed an increase in the average time between injections for the combination vs. the monotherapy groups (177 vs.151 days, p< 0.001). Mean change from baseline BCVA ranged from +0.6 to +3.4 lines in the monotherapy group and +1.3 to +2.8 in the combination group. Mean CFT decrease from baseline was 165–230 µm in the monotherapy group and 136–175 µm in the combination therapy group.
In another study, patients received bevacizumab followed two weeks later by the dexamethasone implant. They were followed monthly and evaluated at each visit for BCVA, CFT and intraocular pressure (IOP).7 When macular edema recurred, they received bevacizumab retreatment. Results demonstrated improvement in visual acuity and prolonged time between injections with the combination therapy compared with either medication on its own
During the study period, 97 percent of participants had an improvement in BCVA with a mean visual acuity improvement of 11 to 25 letters. Participants also experienced a decrease in CFT and the study showed a trend toward stabilization of the reinjection interval at an average of 126 days. This makes the timing of rebound edema and the need for reinjection more predictable.
Reinjection Intervals
| Take-Home Point |
| Combination therapy consisting of sustained-release steroid implants with intravitreal injections of anti-vascular endothelial growth factors has shown promising results in improvement of best-corrected visual acuity and thinning of central foveal thickness in patients with retinal vein occlusions. |
Additionally, we observed no statistically significant difference among six injection cycles, suggesting that the reinjection interval does not change over time. This long-range predictability may play an important role in caring for patients who are significantly burdened by monthly office visits, as it would allow them to safely receive fewer than three cycles of injections per year.9 RS
References
1. Branch Vein Occlusion Study Group. Argon laser photocoagulation for macular edema in branch vein occlusion. Am J Ophthalmology. 1984;98:271–282.2. Glanville J, Patterson J, McCool R, et al. Efficacy and safety of widely used treatments for macular oedema secondary to retinal vein occlusion: a systematic review. BMC Ophthalmol. 2014;14:7.
3. Central Vein Occlusion Study Group. Evaluation of grid pattern photocoagulation for macular edema in central vein occlusion. Ophthalmology. 1995;102:1425–1433.
4. Campochiaro PA, Heier JS, Feiner L, et al. Ranibizumab for macular edema following branch retinal vein occlusion: six-month primary end point results of a phase III study. Ophthalmology. 2010;117:1102–1112.
5. Brown DM, Campochiaro PA, Singh RP, et al. Ranibizumab for macular edema following central retinal vein occlusion: six-month primary end point results of a phase III study. Ophthalmology. 2010;117:1124–1133.
6. Haller JA, Bandello F, Belfort R, et al; Randomized, sham-controlled trial of dexamethasone intravitreal implant in patients with macular edema due to retinal vein occlusion. Ophthalmology. 2010;117:1134–1146.
7. Singer MA, Bell DJ, Woods P, et al. Effect of combination therapy with bevacizumab and dexamethasone intravitreal implant in patients with retinal vein occlusion. Retina. 2012;32:1289-1294.
8. Singer MA, Bell DJ, Woods P, et al. Effect of combination therapy with bevacizumab and dexamethasone intravitreal implant in patients with retinal vein occlusion. Retina. 2012;32:1289-1294.
9. Singer, MA, Tyler L, Woods P, Singer JJ. Long term analysis of combination therapy using anti-vegf agents and Dexamethasone intravitreal implant in retinal vein occlusion. Paper presented at 29th Meeeting of Club Jules Gonin; September 5, 2014; Zurich, Switzerland; and 47th Annual Scientific Meeting of Retina Society, September 14, 2014; Philadelphia, PA.



