Moreover, the mechanism by which ZIKV causes fetal microcephaly is still unknown as well. Reports suggest that the virus is able to evade the normal immunoprotective barrier provided by the placenta,4 and its neurotropic properties directly damage the brain during development. Alternatively, the placental response to the virus is the main cause of the brain damage since the virus can interrupt formation of the outer placenta, which might cause or contribute to microcephaly.4
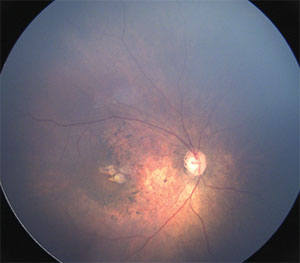 |
| Figure 1. Wide-angle fundus image of the right eye of an infant with congenital Zika syndrome reveals an optic disc hypoplasia, gross macular pigment mottling and juxtafoveal chorioretinal atrophic lesions. |
The virus was first identified in 1947 in a rhesus monkey found in the Zika forest near Kampala, Uganda.1 Five years later, it was isolated in Africans for the first time.5 Then, the virus migrated to the Asian continent during the 1940s as a different strain from the one found in Africa.6
During the past two decades, the Asian strain has been causing outbreaks outside of Asia in other locations such as on Yap Island (Micronesia), in French Polynesia and on Easter Island in Chile.7
However, the most recent and biggest ZIKV outbreak in history started in May 2015 in northeastern Brazil.7 In 2015, an estimated 400,000 to 1.3 million people were infected by ZIKV.7 The recent report of a possible association between ZIKV infection and an epidemic of microcephaly among neonates in Brazil has attracted significant global attention.8
This rapid spread of ZIKV beyond Africa and Asia to the Americas and Europe associated with the novel congenital Zika syndrome outbreak led the World Health Organization to declare this ZIKV epidemic a global public health emergency earlier this year.9
Systemic Manifestations
Only 20 percent of patients infected with ZIKV complain of mild symptoms such as headache, maculopapular rash, arthralgia and conjunctivitis, which usually last for one week.4 Severe disease and fatalities caused by ZIKV were never previously described before the most recent studies from Brazil and French Polynesia, which described a neurotropism of the virus and the increased chance of Guillain-Barré Syndrome and other neurological manifestations.2 Furthermore, microcephaly, hearing loss, limb abnormities and ocular findings were recently described as complications of ZIKV when the infection occurs during pregnancy.2,10–12
 |
The current evidence of ZIKV infection relies on the molecular detection of viral RNA, which is positive only in a brief period of viremia. The currently available serological testing that identifies IgM and IgG antibodies specific for ZIKV is unreliable due to its cross-reactivity with other flaviviruses, and further studies are necessary to better elucidate these findings and their correlation to ZIKV.13 Camila Ventura, MD, and colleagues in Brazil recently published a study in which 40 infants with microcephaly were evaluated.12 They tested using an IgM antibody-captured enzyme-linked immunosorbent assay (MAC-ELISA) of the cerebrospinal fluid in 24 of 40 infants (60 percent). All 24 infants had a positive MAC-ELISA for ZIKV in the cerebrospinal fluid; 14 were from 22 infants (63.3 percent) with ophthalmoscopic findings and 10 were from 18 infants (55.6 percent) without ophthalmoscopic findings.
Retinal Manifestations of ZIKV
A mild course of the disease can include anterior uveitis and a non-purulent conjunctivitis.14 Dr. Ventura and colleagues published the first report of three children with presumed ZIKV congenital infection and ocular abnormalities.15 They identified retinal alterations such as pigment mottling and chorioretinal atrophy in the macular region of the infants.
Further studies in two cities in northeast Brazil, Recife and Salvador, reported similar ocular abnormalities affecting the retina as well as optic disc abnormalities in these infants.10–12 These findings included gross macular pigment mottling, macular chorioretinal atrophy, optic nerve hypoplasia, increased cup-to-disc ratio (Figures 1 and 2), iris coloboma and lens subluxation.
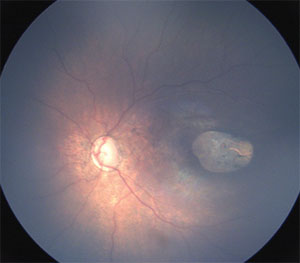 |
| Figure 2. Wide-angle fundus image of the left eye of an infant with congenital Zika syndrome shows optic disc hypoplasia and a sharply demarcated area of chorioretinal atrophy in the macula. |
In a study conducted in Recife, nine of 20 eyes (45 percent) had optic nerve hypoplasia, pallor and increased cup-to-disk ratio.11 The pathophysiology of these lesions in these infants is thought to be related directly to the virus or an associated toxin leading to an inflammatory reaction. In addition, this same mechanism could be responsible for the severe cerebral findings, such as abnormal development and cerebral calcification.
Dr. Ventura and colleagues12 hypothesized that ZIKV may cause more severe ocular abnormalities when the infection occurs in the first or second trimester of pregnancy, as it does in other congenital infections such as toxoplasmosis, rubella and cytomegalovirus. Furthermore, other unknown factors, such as the amount of virus in the circulation and the immunologic response of mother and/or fetus, may play an important role in the formation of these abnormalities in newborns.11
Future Perspectives
Further efforts are needed to understand the pathogenesis of the ocular manifestations, to develop specific antiviral therapy and to facilitate vaccination against ZIKV and other arboviruses. First and foremost, mosquito eradication programs are critical to reduce the infection rates of ZIKV and other mosquito-borne illnesses such as DFV and CHIKV. These efforts are already underway globally and rely primarily on environmental modifications.
Furthermore, genetic manipulation of mosquito populations, such as the recently described gene-drive system that can introduce female sterility into a target vector population, may enhance these efforts.16 RS
REFERENCES
1. Dick GW, Kitchen SF, Haddow AJ. Zika virus. I. Isolations and serological specificity. Trans R Soc Trop Med Hyg. 1952;46:509-520.
2. World Health Organization (WHO). Epidemiological alert: neurological syndrome, congenital malformations, and Zika virus infection. Implications for public health in the Americas. December 1, 2015. Available at: http://www.paho.org/hq/index.php?option=com_docman&task=doc_view&Itemid=270&gid=32405&lang=en. Accessed May 9, 2016.
3. Chan JF, Choi GK, Yip CC, Cheng VC, Yuen KY. Zika fever and congenital Zika syndrome: An unexpected emerging arboviral disease. J Infect. 2016;72:507-524.
4. Adibi JJ, Marques, ET Jr, Cartus A, Beiji RH. Teratogenic effects of the Zika virus and the role of the placenta. Lancet. April 9, 2016. [epub]
5. Dick GW. Zika virus. II. Pathogenicity and physical properties. Trans R Soc Trop Med Hyg. 1952;46:521-534.
6. Pinto Junior VL, Luz K, Parreira R, Ferrinho P. Zika virus: a review to clinicians. Acta Med Port. 2015;28:760–765
7. Zika virus in the Americas: a review for clinicians. Sampathkumar P, Sanchez JL. Mayo Clin Proc. 2016;91:514-521.
8. Schuler-Faccini L, Ribeiro EM, Feitosa IM, et al. Possible association between Zika virus infection and microcephaly—Brazil, 2015. MMWR Morb Mortal Wkly Rep. 2016;65:59-62.
9. World Health Organization (WHO). WHO statement on the first meeting of the International Health Regulations (2005) (IHR 2005) Emergency Committee on Zika virus and observed increase in neurological disorders and neonatal malformations. February 1, 2016. Available at: http://www.who.int/mediacentre/news/statements/2016/1st-emergency-committee-zika/en/. Accessed May 9, 2016.
10. De Paula Freitas B, de Oliveira Dias JR, Prazeres J, et al. Ocular findings in infants with microcephaly associated with presumed Zika virus congenital infection in Salvador, Brazil. JAMA Ophthalmol. February 9, 2016. [Epub ahead of print]
11. Ventura CV, Maia M, Ventura BV, et al. Ophthalmological findings in infants with microcephaly and presumable intra-uterus Zika virus infection. Arq Bras Oftalmol. 2016;79:1-3.
12. Ventura CV, Maia M, Travassos SB, Martins TT, Patriota F, Nunes ME, et al. Risk factors associated with the ophthalmoscopic findings identified in infants with presumed Zika virus congenital infection. JAMA Ophthalmol. May 26, 2016. [Epub ahead of print]
13. Petersen LR, Jamieson DJ, Powers AM, Honein MA. Zika virus. New Engl J Med. 2016;374:1552-1563.
14. Fontes BM. Zika virus-related hypertensive iridocyclitis. Arq Bras Oftalmol. 2016;79:63.
15. Ventura CV, Maia M, Bravo-Filho V, Góis AL, Belfort R Jr. Zika virus in Brazil and macular atrophy in a child with microcephaly. Lancet. January 8, 2016. [Epub]
16. Sikka V, Chattu VK, Popli RK, Galwankar SC, Kelkar D, Sawicki SG, et al. The emergence of Zika virus as a global health security threat: A review and a consensus statement of the INDUSEM Joint Working Group (JWG). J Glob Infect Dis. 2016;8:3–15.




