Yes, the headline is correct. The Centers for Medicare and Medicaid Services (CMS) developed another process to audit and monitor provider claims. In 2014, CMS initiated a pilot program called Targeted Probe and Education, or TPE. Due to its success and “favorable outcomes,” TPE is being rolled out to all Medicare Administrative Contractors (MACs). On September 15, 2017, Change Request 10249 expanded the TPE program to include all MACs effective October 1 last year.1 According to CMS, “The process is only used with those who have high denial rates or unusual billing practices.”2
Targeted Topics
During the pilot program, CMS chose the topics for review. Now, the MACs will be choosing the topics under review. As
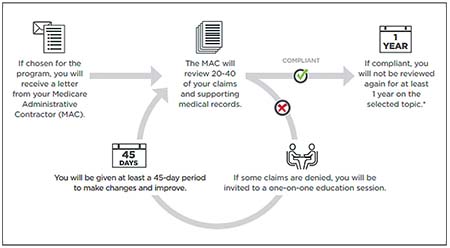 |
| How the Targeted Probe and Education review process works. A provider gets three rounds to be compliant to avoid further action. (Image provided by Centers for Medicare and Medicaid Services.) |
of this writing, only one MAC, Novitas Solutions Inc., which covers 11 states and the District of Columbia, has published a list of topics for review.3 The list includes some services like new and established Evaluation and Management (E/M) office visit codes (99201 to 99205 and 99211 to 99215). More specific to the subspecialty of retina, two high-volume drugs are on the list:
J0178 – Injection, aflibercept, 1 mg; and
J2778 – Injection, ranibizumab, 0.1 mg.
Novitas—not to be confused with Novartis—provides key points regarding injections and billing for drugs.4 The following topics are listed for injection services:
Where the sole purpose of an office visit was for patient to receive an injection, payment may be made only for the injection service (if it is covered).
Pay separately for those injection services only if no other physician fee schedule service is being paid.
The drug is separately payable.
All injection claims must include specific name of the drug and dosage.
Novitas further conditions that drugs and biologicals are covered if all requirements are met:
Not usually self-administered by patients.
Meet all general requirements for coverage of items as incident to a physician’s services.
Reasonable and necessary for diagnosis or treatment of illness or injury for which they are administered according to accepted standards of medical practice.
Not excluded as immunizations.
Have not been determined by Food and Drug Administration (FDA) to be less than effective.
Novitas lists the TPE “start date” for new patient E/M codes to begin June 2018, established E/M codes to begin in August, and the TPE process for drug injections to begin in May and June.
The TPE Process
A targeted review of providers with high denial rates or unusual billing practices determines who will be audited. As subspecialists, retinal specialists, when compared to all of ophthalmology, are outliers. Being an outlier does not mean you are doing something wrong; it only means subspecialists have different claims and billing patterns.
If the TPE process targets a clinician, the MAC will send a letter to the provider asking for supporting documentation for 20 to 40 claims. After submission, the auditor for the MAC will review the supporting documentation and the MAC will notify the clinician of the outcome of the review.
If the claims are compliant, the process likely ends for the targeted topic for at least one year. However, if claims are denied or the audit identifies issues, the clinician gets a “one-on-one” education session. Following the education session, the provider is given 45 days to make changes and improve. After the 45 days the process starts over and the MAC reviewer requests another sample of 20 to 40 claims, but for dates of service after the one-on-one session.5
If the practice is still non-compliant or unsuccessful after three rounds and fails to show adequate improvement, the provider will be referred to the CMS for the next steps. They may include 100 percent prepay review, extrapolation, referral to a recovery auditor or other action.6
 |
All of this increased scrutiny can create significant problems for the practice.
Following each round, the provider receives a letter that details the results of the review. Unfortunately, the acceptable error rates are not published and may vary by MAC. The letter may arrive before or after the one-on-one education.7
Each provider has three rounds to show compliance to avoid further action beyond TPE. It is also possible that a clinician could be involved in more than one TPE at a time. For example, a request for E/M and ranibizumab (Lucentis, Roche/Genentech) claims could occur concurrently.
Monitor Your MAC for Updates
CMS says the TPE process will reduce the number of providers receiving audit requests. The agency’s goal is “to help you quickly improve.” During the pilot program, we received a few calls from clients submitting documentation. Each client submitted what was requested, and we have yet to hear of anyone subjected to more than one or two rounds in the pilot TPE process.
The TPE rollout is new and therefore it’s too early to know how each MAC will handle it. Watch for any TPE requests and do your best to submit thorough documentation supporting any claims in the request. For additional information, monitor your MAC for any MAC-specific updates. RS
Mr. Mack is a senior consultant with Corcoran Consulting Group. He can be reached at 1-800-399-6565 or at www.corcoranccg.com.
REFERENCES
1. Department of Health and Human Services. Centers for Medicare & Medicaid Services. Change Request 10249. September 15, 2017. Available at: https://www.cms.gov/Regulations-and-Guidance/Guidance/Transmittals/2017Downloads/R1919OTN.pdf. Accessed May 27, 2018.
2. Centers for Medicare & Medicaid Services. Improving the Medicare claims review process. Available at: https://www.cms.gov/Research-Statistics-Data-and-Systems/Monitoring-Programs/Medicare-FFS-Compliance-Programs/Medical-Review/Downloads/What_Is_TPE-Infosheet.pdf. Accessed May 27, 2018.
3. Novitas Solutions Inc. Medicare Administrative Contractor. Targeted Probe and Educate. Topics for review. Available at: https://www.novitas-solutions.com/webcenter/portal/MedicareJL/pagebyid;jsessionid=aqn63fElmDDqQ1kQCATd8DQlWUp7zRpRo5S-ncPlW_779cHjFZv9!435923927!-1075561717?centerWidth=100%25&contentId=00161300&leftWidth=0%25&rightWidth=0%25&showFooter=false&showHeader=false&_adf.ctrl-state=qye1z877y_4&_afrLoop=787493541774661#!%40%40%3F_afrLoop%3D787493541774661%26centerWidth%3D100%2525%26contentId%3D00161300%26leftWidth%3D0%2525%26rightWidth%3D0%2525%26showFooter%3Dfalse%26showHeader%3Dfalse%26_adf.ctrl-state%3Dimz51zvdg_4. Accessed May 27, 2018.
4. Novitas Solutions Inc. Medicare Administrative Contractor. Targeted Probe and Educate (TPE) injections–drugs and biologicals. Slide presentation. Available at: https://www.novitas-solutions.com/webcenter/content/conn/UCM_Repository/uuid/dDocName:00176701. Accessed May 27, 2018.
5. Centers for Medicare & Medicaid Services. Improving the Medicare claims review process. Available at: https://www.cms.gov/Research-Statistics-Data-and-Systems/Monitoring-Programs/Medicare-FFS-Compliance-Programs/Medical-Review/Downloads/What_Is_TPE-Infosheet.pdf. Accessed May 27, 2018.
6. Centers for Medicare & Medicaid Services. Targeted Probe and Educate (TPE). What if my accuracy still doesn’t improve? Available at: https://www.cms.gov/Research-Statistics-Data-and-Systems/Monitoring-Programs/Medicare-FFS-Compliance-Programs/Medical-Review/Targeted-Probe-and-EducateTPE.html#probe. Accessed May 27, 2018.
7. Targeted Probe and Educate (TPEs) Q & A’s. Available at: https://www.cms.gov/Research-Statistics-Data-and-Systems/Monitoring-Programs/Medicare-FFS-Compliance-Programs/Medical-Review/Downloads/TPE-QAs.pdf. Accessed May 27, 2018.



