 |
|
Bios
Dr. Felfeli is an ophthalmology resident at the University of Toronto. Mr. Papanikolaou is a research student at University Health Network, Toronto. Dr. Mandelcorn is an associate professor of ophthalmology at the University of Toronto. DISCLOSURES: The authors have no relevant financial relationships to disclose. |
We previously described the macular hole hydrodissection technique for closure of large, persistent and recurrent macular holes.1 This technique is effective in closing up to 87 percent of the most challenging macular holes, with 95 percent of patients experiencing vision improvement.1 Despite the success of this technique, in our experience, for macular holes that are exceedingly large (>800 µm), a graft such as a human amniotic membrane (hAM) is often necessary to ensure complete closure.
Initial uses of hAM in ophthalmology
The amniotic membrane is the innermost layer of fetal membranes, with cells that possess a number of qualities suitable for use in regenerative medicine, such as low immunogenicity, promotion of epithelization and anti-inflammatory properties.2 One of the earliest uses of hAM in ophthalmology was reported in 1940 by A. de Rötth, MD, for repair of conjunctival defects and reconstruction.3 Subretinal implantation of the hAM was found to be tolerated in rabbits as Philip Rosenfeld, MD, and colleagues described in 1999.4 RPE cells cultured on hAM have also been shown to maintain retinal homeostasis and improve eye function.5
Use of hAM for macular holes
It’s been suggested that hAM, in part because of its anti-inflammatory and pro-healing tendencies, serves as a highly effective biologic structural scaffold to promote centripetal migration of macular hole edges and resultant closure.6 In 2018, a small case series of eight recurrent macular holes by Stanislao Rizzo, MD, and colleagues in Italy demonstrated a 100-percent closure rate with the use of hAM.7,8 Studies have also shown partial recovery of macular sensitivity with microperimetric testing at the edges of the plug, with significant improvement in best-corrected visual acuity.8
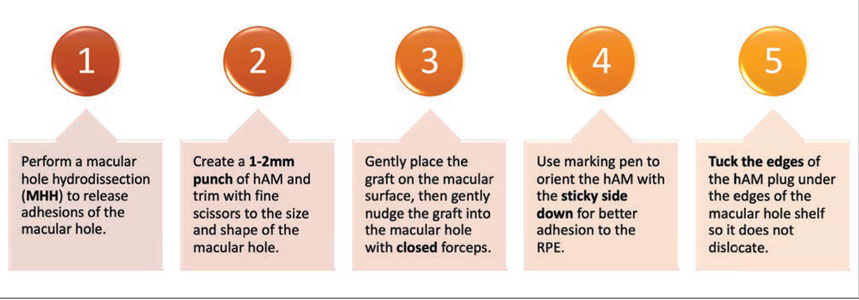 |
| Figure 1. The five key steps in our approach to macular hole repair with human amniotic membrane. |
Our approach to MH repair with hAM
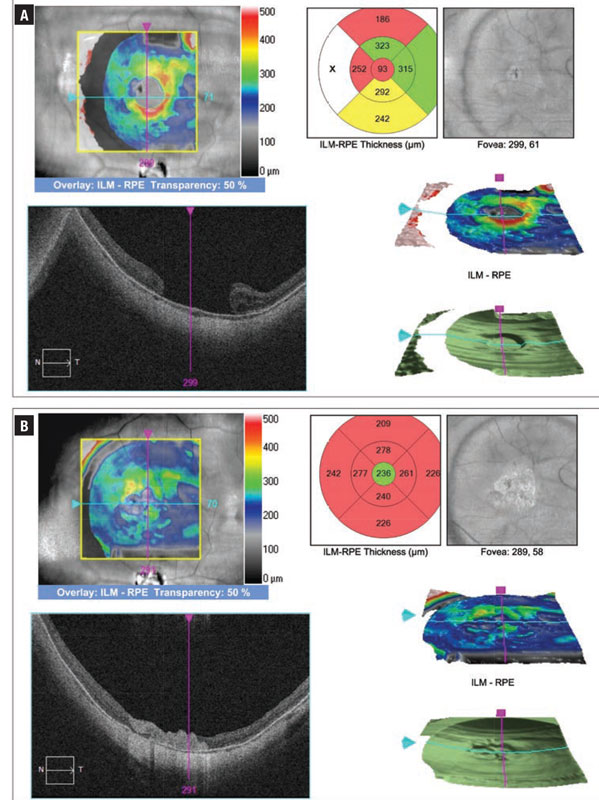 |
| Figure 2. Optical coherence tomography macular cube scans before (A) and after (B) macular hole repair with the macular hole hydrodissection technique and human amniotic membrane. |
There are a few critical steps in hAM macular hole surgery that are important to consider when placing a hAM in a macular hole (Figure 1). First, one of the most important steps is to ensure that any adhesions of the macular hole to the underlying RPE are well-separated. This can be achieved with the macular hole hydrodissection (MHH) technique by manipulating the edges of the macular hole with a soft-tipped cannula with proportional reflux, followed by gentle passive aspiration.1 This process creates a shelf between the macular hole edges and the retinal pigment epithelium, under which the hAM can be placed.
Next, we use a 1- to 2-mm punch to fashion the amniotic membrane graft. We often trim this with fine scissors to a size just a little larger than the macular hole so the graft will fit nicely in the macular hole. It’s important that the diameter of the plug almost matches the macular hole, because plugs smaller than the macular hole won’t attach under the shelf of the macular hole edges, and larger plugs won’t fit in the macular hole or create folds from tissue redundancy.
We then place the hAM in the vitreous cavity with a fine gripping forceps and tuck it into the space created around the macular hole edges. It’s helpful to gently place the graft on the macular surface so it doesn’t float away, and then nudge the graft into the hole with the closed forceps so it doesn’t stick to the teeth of the forceps.
It’s also important to place the graft with the stromal layer side down (it’s sticky and non-shiny) so it will adhere to the RPE once it’s in the macular hole. We often mark the non-sticky side with a small dot using a marking pen so we can identify it once it’s placed in the vitreous cavity. The plug edges are tucked under the edges of the macular hole shelf. This is a crucial step to ensure the hAM graft doesn’t dislocate postoperatively. A fluid-air exchange is carried out and the eye is then flushed with SF6 gas (20%).
A case of MH repair with hAM
The accompanying video shows a case of a 52-year-old woman with a history of high myopia and surgical repair for a macular hole-related macular detachment with a persistent open MH. She had been followed for four years and decided to undergo secondary repair of her macular hole that was measured preoperatively at 1,458 µm with a visual acuity of 20/400 (Figure 2). She underwent MHH with a hAM plug and had an excellent anatomical closure of the macular hole. Her postoperative visual acuity recovered to 20/70 at seven months.
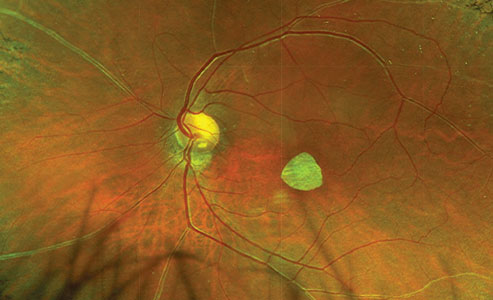 |
| Figure 3. Widefield color fundus photography depicts a well-positioned round-shaped human amniotic membrane plug in the macular area following macular hole repair. |
Figure 3 depicts another case with a well-positioned round-shaped hAM plug as shown on a color fundus image.
Alternative techniques
Alternative techniques using lens capsular flap, autologous transplantation of the internal limiting membrane and neurosensory retinal free flap, and autologous blood have been reported, but with associated intraoperative and postoperative complications.10 Similarly, neurosensory retinal free flap transplantation can be challenging; the most appropriate size of the neurosensory free flap is unclear, as it may contract after being implanted into the macular hole.11
Bottom line
Overall, we think the use of hAM plugs is a promising new way to treat large macular holes. When combined with MHH, this technique may offer good anatomical closure rates and visual recovery. RS
REFERENCES
1. Felfeli T, Mandelcorn ED. Macular hole hydrodissection: Surgical technique for the treatment of persistent, chronic, and large macular holes. Retina. 2019;39:743-752.
2. Ramuta TZ, Kreft ME. Human amniotic membrane and amniotic membrane–derived cells: How far are we from their use in regenerative and reconstructive urology? Cell Transplant. 2018;27:77-92.
3. de Rötth A. Plastic repair of conjunctival defects with fetal membranes. Arch Ophthalmol. 1940;23:522-525.
4. Rosenfeld P, Merritt J, Hernandez E, Meller D, Rosa RJ, Tseng S. Subretinal implantation of human amniotic membrane: A rabbit model for the replacement of Bruch’s membrane during submacular surgery. Invest Ophthalmol Vis Sci. 1999;40:206.
5. Ohno-Matsui K, Ichinose S, Nakahama KI, et al. The effects of amniotic membrane on retinal pigment epithelial cell differentiation. Mol Vis. 2005;11:1-10.
6. Malhotra C, Jain AK. Human amniotic membrane transplantation: Different modalities of its use in ophthalmology. World J Transplant. 2014;4:111-121.
7. Rizzo S, Caporossi T, Tartaro R, et al. A human amniotic membrane plug to promote retinal breaks repair and recurrent macular hole closure. Retina. 2019;39:S95-S103.
8. Caporossi T, Pacini B, Bacherini D, Barca F, Faraldi F, Rizzo S. Human amniotic membrane plug to promote failed macular hole closure. Sci Rep. 2020;10:1-8.
9. Tsai DC, Huang YH, Chen SJ. Parafoveal atrophy after human amniotic membrane graft for macular hole in patients with high myopia. Br J Ophthalmol. 2020:1-9.
10. Chen S-N, Yang C-M. Lens capsular flap transplantation in the management of refractory macular hole from multiple etiologies. Retina. 2016;36:163-170.
11. Liu PK, Chang YC, Wu WC. Management of refractory macular hole with blood and gas-assisted autologous neurosensory retinal free flap transplantation: A case report. BMC Ophthalmol. 2018;18:18-21.




