| ABOUT THE AUTHOR | |
 | Dr. Isom is in private practice in Salt Lake City, Utah and participates as adjunct faculty at the Moran Eye Center. DISCLOSURES: Dr. Isom has no relevant conflicts to disclose. |
Surgical interventions began in the 1930s with the introduction of the scleral buckle, followed by pneumatic retinopexy and, more recently, vitrectomy with or without a scleral buckle. This article will discuss various methods of subretinal fluid drainage with case presentations of non-lasered drainage retinotomies.
Methods of Subretinal Fluid Drainage
Various techniques exist for draining subretinal fluid during vitrectomy for repair of rhegmatogenous retinal detachments. Three common techniques include:• Drainage from a preexisting retinal break.
• Perfluorocarbon liquids.
• Draining retinotomies.
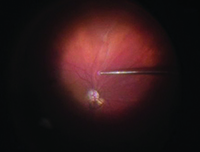
of inferior non-lasered drainage
retinotomy of a macula-off retinal
detachment.
Video available at http://goo.gl/7xdexW.
Ideally fluid is drained from an already present retinal break during a fluid-air exchange with complete flattening of the retina. This technique avoids the need to create additional retinal holes and the cost and possible complications of perfluorocarbon liquids.
Draining through a peripheral break can be challenging due to residual vitreous around the break, difficulty of access in phakic eyes and residual subretinal fluid. Significant amounts of residual subretinal fluid can lead to retinal folds, which have been reported in up to 3 percent of cases postoperatively in one series.1
Intraoperative head tilt can minimize residual, posterior subretinal fluid. This makes the area of the break the most dependent to pool the fluid closer to the hole. Although the head tilt may compromise the view, a greater amount of fluid may be removed to decrease the risk of postoperative retinal folds.
Role of Liquid Fluorocarbons
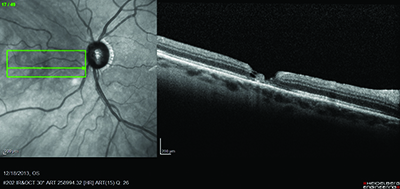 |
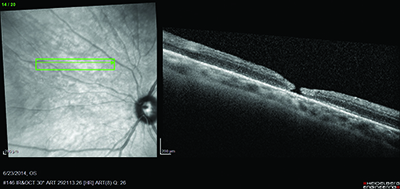 |
| Figure 2. Postoperative optical coherence tomography shows no associated subretinal fluid or redetachment from the retinotomy. |
Some surgeons prefer to use liquid fluorocarbons routinely to avoid the need for posterior drainage retinotomies. Although perfluorocarbons have been a useful addition to the repair of retinal detachments, they remain expensive and carry the risk of retention in the subretinal space.
One study found the rate of subretinal perfluorocarbon to be just over 11 percent.3 However, all of the cases of retained subretinal perfluorocarbon involved retinectomies of over 120°. Valved cannulas and a greater familiarity with these liquids can decrease the risk of subretinal retention.
However, as surgeons’ outcomes are being more closely scrutinized for their cost-effectiveness, the cost of items such as perfluorocarbons may begin to affect their use in
routine cases.
Drainage Retinotomies
Steve Charles, MD, first popularized drainage retinotomies to aid in the transretinal subretinal fluid drainage during a gas exchange. A more complete drainage of subretinal fluid is usually achieved because the surgeon can drain from the “bottom of the bowl” as opposed to draining from a peripheral retinal break. A more complete drainage of subretinal fluid decreases the risk of postoperative retinal folds.The complications of posterior drainage retinotomies include visual field scotomas, risk of subsequent epiretinal membrane and proliferative vitreoretinopathy, as well as subretinal choroidal neovascular membrane formation.
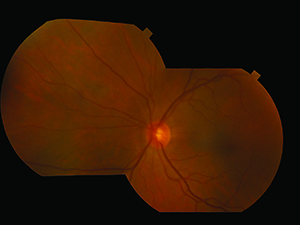 |
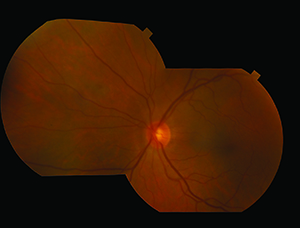 |
| Figure 3. Fundus photographs show an almost normal posterior pole in the area of the retinotomy. |
Non-lasered Retinotomies
The rationale to laser a drainage retinotomy is to prevent redetachment from the retinotomy site. However, a hole in the retina does not lead to a detachment unless another force is acting upon it. The retina “wants” to stay attached through a variety of systems: the negative pressure gradient between the vitreous and the choroid; the pumping mechanism of retinal pigment epithelium; and subcellular “glue” components of the retinal pigment epithelium. This is evidenced by macular holes that are rarely the cause of rhegmatogenous retinal detachments, aside from in very myopic eyes.The easiest way to create a drainage retinotomy is with diathermy. We have found that cauterizing the retina briefly with diathermy to create a non-perforated white spot, followed by perforation with the sharp tip of the diathermy, can create a very small opening through which subretinal fluid may be removed during a gas exchange. Prior removal of all viscous subretinal fluid through a peripheral break will further reduce the expansion of a retinotomy during a fluid-gas exchange.
In uncomplicated rhegmatogenous retinal detachment cases, these drainage retinotomies may be left unlasered to decrease the size of the scotoma. It is our experience that patients do not appreciate any scotoma from these small retinotomies, although we have not performed extensive postoperative visual field testing.
| Take-home Point The techniques employed to repair retinal detachments continue to vary and improve over time. Drainage of subretinal fluid from a primary break is ideal, but in cases of persistent posterior fluid, a drainage retinotomy may decrease the risk of postoperative retinal folds and decrease the cost of surgery. These retinotomies may be left unlasered in most routine cases. Further evaluation in a larger series is necessary to evaluate any increase in epiretinal membranes, size of induced visual field scotoma and effect on redetachment rate. |
Surgeons should use caution in using drainage retinotomies in young patients or in cases of inflammation or trauma due to concern for promoting PVR. If there is intraoperative or postoperative concern for PVR, drainage retinotomies should be lasered to prevent redetachment from the site. RS
References
1. Van Meurs JC, Humalda D, Mertens DA, Peperkamp E: Retinal folds through the macula. Doc Ophthalmol. 1991;78:335–340.2. Chang S. Low viscosity liquid fluorochemicals in vitreous surgery. Am J Ophthalmol. 1987;103:38-43.
3. Garcia-Valenzuela E, Ito Y, Abrams GW. Risk factors for retention of subretinal perfluorocarbon liquid in vitreoretinal surgery. Retina. 2004;24:746-752.
4. Richards SC, Maberley AL. Complications of retinotomies for subretinal fluid drainage. Can J Ophthalmol. 1993;28:24-27.



