 |
 |
 |
Corticosteroids are often the primary treatment for noninfectious uveitis, administered via either the topical, regional or systemic routes. Many retina specialists are utilizing intravitreal corticosteroids as adjuncts to anti-VEGF injections in diabetic macular edema and cystoid macular edema secondary to retinal vein occlusions.1
Regional steroids in noninfectious uveitis can effectively treat intraocular inflammation and uveitic macular edema, but their use is perhaps more nuanced and less algorithmic than in other conditions. We outline some guidelines for retina specialists treating noninfectious uveitis to help optimize safety and efficacy while reducing complications, and then we review the available and investigative therapies.
Ensuring the correct diagnosis
Following a careful targeted history and anterior segment and fundus examinations, ocular imaging with optical coherence tomography and angiography will help to classify the primary site of inflammation and can aid in making a diagnosis. Retina specialists should employ a tailored laboratory evaluation for possible systemic inflammatory and especially infectious laboratory workup in all cases of posterior segment-involving uveitis.
If the diagnosis is unclear or if the prescribed treatment has not produced the expected outcome, referral to a uveitis specialist can help direct care,2 particularly when considering systemic treatment with immunosuppressive or biologic therapy (i.e., anti-tumor necrosis factor agents). The use of systemic therapy is beyond the scope of this targeted review.
Starting topical corticosteroids
Topical corticosteroids such as prednisolone acetate can usually be initiated early in the course of uveitis, and can quickly be tapered or discontinued if necessary. Infectious causes should be concomitantly treated with appropriate antibiotic, antiviral, antifungal or antiparasitic therapy.
Prednisolone acetate and similar topical corticosteroids will not penetrate the posterior segment in adequate concentrations to resolve vitreous inflammation, so these are typically insufficient as the primary therapy for intermediate or posterior uveitis. Fluorinated topical steroids such as difluprednate acetate (Durezol, Novartis)3 allow for better penetration into the posterior segment and can be very effective at improving vitreous cells, vitreous haze, and mild uveitic macular edema (UME). These potent corticosteroids also have an increased risk of steroid-induced intraocular pressure and cataract progression, so use them with caution.
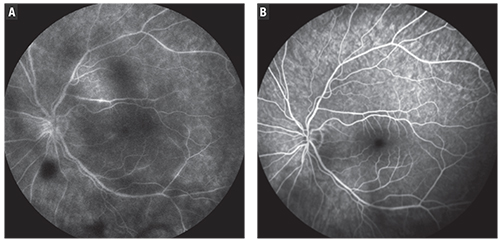 |
| Figure. Fluorescein angiograms show vascular leakage before sub-Tenon’s injection of Kenalog (A) and resolution of much of the leakage after injection (B). |
Transitioning to regional corticosteroids
If topical corticosteroids are insufficient to improve inflammation, or if structural complications such as UME arise, regional corticosteroids can be useful adjunctive or primary treatments.
Again, give consideration to appropriate anti-infective treatments if necessary, because regional steroids can worsen an infection not treated completely, and they are more difficult to discontinue once administered.
The next steps in transitioning to regional corticosteroids are:
• determining the location (periocular vs. intraocular); and
• the desired duration of effect (from months to potentially years).
Periocular corticosteroids
Triamcinolone acetonide (Kenalog, Ranbaxy Laboratories) delivered into the sub-Tenon’s space or through the orbital septum is a readily available, cost-effective option for treating noninfectious uveitis. The most common concentration is 40 mg/ mL (i.e., Kenalog-40). It’s typically delivered in 0.5-mL volumes.
Sub-Tenon’s triamcinolone would typically have a duration of two to three months, and the location provides a depot for slow release of steroid over this interval. Because the crystal sizes are variable in Kenalog suspensions, the duration of action may not be identical even if both eyes are treated from the same vial. Complications can include ptosis, ocular hypertension and cataract.
 |
Intravitreal steroids: short- vs. long-acting
The decision to use an intravitreal steroid may follow administration of a periocular steroid, or if it’s indicated to rapidly resolve intermediate or posterior uveitis with associated UME. Triesence (Alcon),4 preservative-free triamcinolone acetonide, is a single-use injectable suspension designed for intraocular use. While the concentration is similar to its preserved counterpart (40 mg/mL), a typical intravitreal injectable dose would be 4 mg/0.1 mL. It’s usually administered inferotemporally.
Check IOP immediately after injecting Triesence because the medication can produce a sudden spike. Because the crystals in Triesence are uniformly sized, it has a predictable duration of action of roughly four to six weeks. The one caveat is that Triesence crystals may clear more rapidly out of a vitrectomized eye than in an eye with intact vitreous.
Retisert
This sutured, surgically implanted fluocinolone implant (Bausch + Lomb) has been in use since 2005 and has been extensively studied. Most recently, the seven-year follow-up data of the MUST trial revealed that the implant performed as well as the systemic therapy over the first five years.5 However, the implant has also had significant local complications, including the need for cataract surgery in most patients and glaucoma surgery in up to 40 percent of patients.
Ozurdex
Ozurdex (dexamethasone intravitreal 0.7-mg implant, Allergan) is approved for noninfectious uveitis.6 Retina specialists may comfortably incorporate Ozurdex injections because they are routinely used for diabetic macular edema and retinal edema secondary to retinal vein occlusions along with anti-VEGF injections.
Ozurdex injections are given via a beveled entry into the sclera with a 25-gauge injector system. IOP rise predictably occurred at four to six weeks in most of the clinical studies, so this provides a convenient time frame in which to see a patient in follow-up. Avoid these injections if the posterior capsule had been disrupted in cataract surgery, but they can be safely used in patients who have had a YAG capsulotomy.
The duration of action of Ozurdex is typically three to four months. The POINT trial,7 a recent prospective randomized comparative study of intravitreal triamcinolone, Ozurdex and sub-Tenon’s triamcinolone for UME, determined both intravitreal injections had a greater and faster therapeutic effect compared with sub-Tenon’s injections, but found no significant differences between the two intravitreal injections.
 |
Yutiq
Yutiq (0.18-mg fluocinolone acetonide intravitreal implant, EyePoint Pharmaceuticals) received Food and Drug Administration approval in October 2018 for the treatment of noninfectious posterior segment uveitis.
This implant is virtually identical to the Iluvien implant (0.19 mg fluocinolone acetonide, Alimera Sciences) approved for the treatment of diabetic macular edema in the United States and Europe, and for noninfectious uveitis in Europe.8
Yutiq releases approximately one-third the dose of Retisert over roughly the same duration—three years.
A one-year confirmatory study of the ability of the Yutiq implant to reduce uveitis recurrences showed that, compared to sham injections, Yutiq-treated patients were roughly half as likely to experience recurrence of posterior uveitis requiring steroid rescue or systemic treatment at one year—32.7 percent in fluocinolone acetonide treated eyes vs. 59.6 percent in sham-treated eyes.9
Suprachoroidal triamcinolone
A novel development from Clearside Biomedical is Xipere, previously known as CLS-TA, a proprietary delivery system of triamcinolone acetonide into the suprachoroidal space.
The Phase III PEACHTREE trial evaluated the company’s suprachoroidal triamcinolone acetonide platform in patients with macular edema secondary to noninfectious uveitis.10 The study showed that significantly more patients gained 15 letters in the Xipere-treated group (47 percent) compared with controls (16 percent) at six months (p< 0.001).
The CLS-TA group also had a 50-percent reduction of macular edema, and the vast majority (85 percent) didn’t require rescue therapy during the six-month study period. This may one day provide an approved delivery system for uniform periocular corticosteroid in noninfectious uveitis.
Bottom line
The armamentarium of corticosteroid injections will likely broaden over the next few years as the longer-acting fluocinolone implants (Iluvien, Yutiq) and the novel delivery systems (Xipere) become more widely available. Retina specialists can use steroid injections that we utilize for other conditions and apply that experience to noninfectious uveitis. Consultation with a uveitis specialist can help guide systemic therapy and give insight into using regional corticosteroid injections into care for these patients. RS
REFERENCES
1. American Society of Retina Specialists (ASRS) 2018 Preferences and Trends (PAT) Survey. July 2018. https://www. asrs.org/clinical/pat-survey
2. American Uveitis Society. Available at: http://www. uveitissociety.org
3. Durezol [package insert]. Fort Worth, TX: Alcon Laboratories; 2017.
4. Triesence. [package insert]. Fort Worth, TX: Alcon Laboratories; 2014.
5. Writing Committee for the Multicenter Uveitis Steroid Treatment (MUST) Trial and Follow-up Study Research Group, Kempen JH, Altaweel MM, Holbrook JT, Sugar EA, Thorne JE, Jabs DA. Association between long-lasting intravitreous fluocinolone acetonide implant vs systemic anti-inflammatory therapy and visual acuity at 7 years among patients with intermediate, posterior, or panuveitis. JAMA. 2017;317:1993-2005.
6. Ozurdex [package insert]. Madison, NJ: Allergan; 2018.
7. Multicenter Uveitis Steroid Treatment Trial Research Group; Writing Committee: Thorne JE, Sugar EA, Holbrook JT, et al. Periocular triamcinolone vs. intravitreal triamcinolone vs. intravitreal dexamethasone implant for the treatment of uveitic macular edema: The PeriOcular vs. INTravitreal corticosteroids for uveitic macular edema (POINT) Trial. Ophthalmology. 2018 September 20. [Epub ahead of print]
8. Alimera Sciences receives acceptance of filing to obtain approval for Iluvien uveitis indication in Europe. [press release] Atlanta, GA: Alimera Sciences; January 4, 2018. https://investor. alimerasciences.com/news-releases/news-release-details/ alimera-sciences-receives-acceptance-filing-obtain-approval. Accessed December 13, 2018.
9. Jaffe GJ. Confirmatory 1-year study results of an injectable fluocinolone acetonide intravitreal insert to treat noninfectious posterior segment uveitis. Paper presented at American Society of Retina Specialists Annual Meeting: Vancouver; July 25, 2018. 10. Yeh S. Suprachoroidally injected CLS-TA improves visual acuity and macular edema in noninfectious uveitis: Results of the Phase 3 PEACHTREE Study. Paper presented at American Society of Retina Specialists Annual Meeting: Vancouver; July 25, 2018.



