When we encounter a tractional retinal detachment in a patient with diabetes, we must consider if and when to proceed with surgery. Here, we explain our technique.
 |
Performing macular optical coherence tomography is invaluable in assessing the foveal status and membrane extent. Ongoing OCT imaging during follow-up is efficient and can detect TRD progression. Extramacular/extrafoveal TRDs can be observed closely because only about 15 percent of cases progress to the macula in one year, and 21 to 24 percent do so in two years.1 However, very close monitoring is vital because even transient macular detachments may result in permanent visual loss,2,3 and progression to a combined tractional-rhegmatogenous retinal detachment will reduce surgical success rates and visual outcomes.4
Click here to watch a video that shows before and after images of Dr. Schechet’s and Dr. Skondra’s operative approach.
Planning the Operation
When deciding upon surgery, the surgeon must consider the status of the fellow eye, paying particular attention to potentially progressive TRD. In eyes with diabetic TRDs that have undergone vitrectomy, approximately one-third of fellow eyes will also require a vitrectomy for worsened TRD by three years.5 Finally, while considering and discussing with the patient any potential surgery, it’s important to convey the realistic goals and expected outcomes, including risks of complications. While an operation may be technically and anatomically successful with retinal reattachment, preexisting macular ischemia may prevent vision from improving postoperatively.
• Low threshold for pre-
vitrectomy cataract surgery. A good surgical view is crucial for these complex cases, given that close peeling to the retina is necessary. Thus, we have a low threshold for cataract surgery prior to vitrectomy, and we try to avoid combined phacoemulsification/vitrectomy for long, complex TRD cases. We ask the cataract surgeon to make a large capsulorhexis, place a three-piece intraocular lens in the bag to ensure stability under air and with gas tamponade, do a thorough posterior capsule polish and suture the main wound.
• Evaluate systemic status. It’s important to assess and optimize the systemic status of these often-ill diabetic patients before surgery. The mean survival from time of TRD diagnosis is 2.7 years with a 48.7-percent mortality rate at 10 years.6 These long and complex surgeries frequently require general anesthesia, so preoperative clearance from the primary-
care provider and anesthesia are necessary. Also, pre- and perioperative control of diabetes and hypertension decreases the risk of ocular and systemic complications.
• Preoperative PRP, bevacizumab. Planned preoperative panretinal photocoagulation and intravitreal bevacizumab (Avastin, Roche/Genentech) can aid in surgical success. Applying PRP in an attached peripheral retina a few weeks before surgery is helpful as it prevents TRD progression and vascularity, decreases operative time and reduces postoperative inflammation.
We also routinely use intravitreal bevacizumab two to four days before surgery to minimize intraoperative bleeding. One should be prudent and first obtain preoperative medical clearance. A meta-analysis found that pretreatment with anti-VEGF was associated with decreased intraoperative bleeding, iatrogenic retinal breaks, silicone- oil use and need for relaxing retinotomies.7 It has also been shown to reduce operative times.8,9
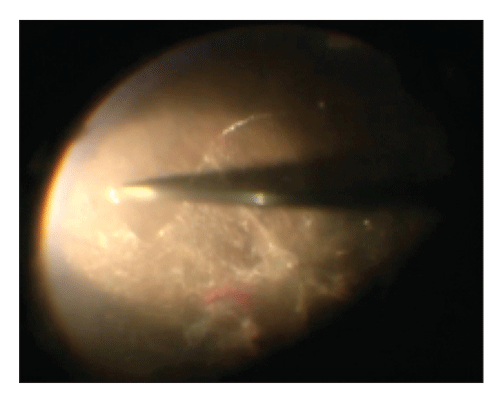 |
| Figure 1. Repeated staining of residualhyaloid with triamcinolone after membrane peeling helps to visualize any residual hyaloid remaining after peeling. |
Intraoperative Steps
• Location of the port. Small-gauge vitrectomy (23-, 25- or 27-gauge) is now the norm for diabetic TRD repair. The proximity of the port to the tip allows better maneuverability and cutting ability of these very adherent fibrovascular membranes (FVMs) to the retina. A recently described hybrid system works very well using a 27-ga vitrector handpiece with 23-ga sclerotomies.10
• Maintain a clear view. Maintaining a clear view during the case is vital. We apply 50% dextrose on the cornea followed by a layer of viscoelastic. We find this keeps the cornea clear much longer than using conventional viscoelastic alone. Contemporary wide-angle,
noncontact viewing systems provide excellent peripheral visualization. A high-magnification macular contact lens can be useful when performing delicate macular
peeling.
• Triamcinolone to visualize FVM dissection. Following the core vitrectomy and releasing 360 degrees of anterior-posterior vitreous traction, attention is directed to the posterior pole. Inducing a posterior vitreous detachment (PVD) is almost always impossible due to extensive and tightly adherent FVMs, so the posterior hyaloid is peeled and separated during FVM dissection and can be visualized better with triamcinolone staining. If you don’t look for residual hyaloid, you won’t find it. So, near the end of the surgery, after membrane peeling, we recommend repeat staining of any potential residual vitreous with triamcinolone (Figure 1). Residual hyaloid left behind postoperatively can contract and be a scaffold for FVM reproliferation.
 |
• Removal of FVMs. Patience and persistence are required for meticulous removal of FVMs. The ultimate goal is to relieve all tractional forces from the retina using techniques such as segmentation and delamination11 and “lift and shave.”12 Leaving residual pegs of fibrovascular tissue is usually acceptable as long as the associated traction is relieved. Sometimes FVMs are too adherent and inseparable from the retina and traction cannot be released, so a focal retinectomy can be considered.
• Finding safe planes to begin FVM dissection. Preoperative planning comes into play here. Analyzing the OCT may aid in finding safe potential planes to begin the FVM dissection. Starting by the optic nerve, move in an “inside-out” approach. Gently perform dissection of the fibrovascular tissue from the nerve.
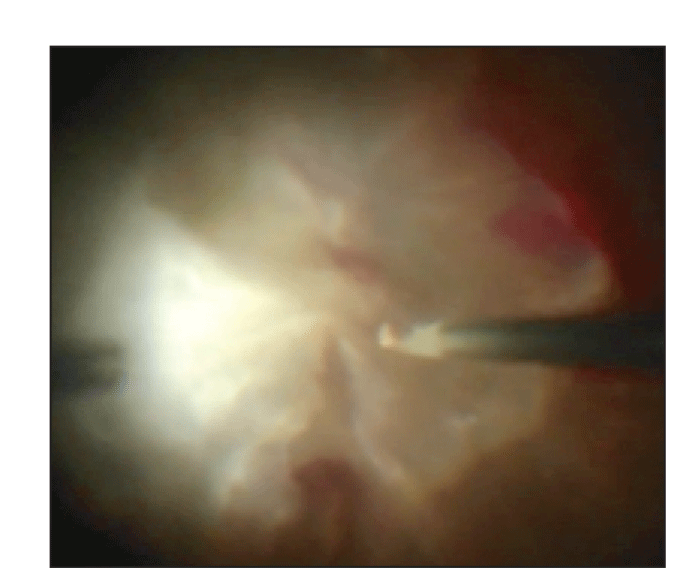 |
| Figure 2. Vertical pneumatic membrane peeler-cutter scissors aid in the dissection of the fibrovascular membrane. |
• Segmentation and delamination. Once a plane is created, carry out segmentation and delamination with utmost care. Sometimes a bimanual approach, using lighting from a chandelier or lighted instruments, is helpful. Often more tools, in addition to the light and cutter, are needed. These can include the delaminating blunt spatula, internal limiting membrane forceps, flex loop and curved horizontal or vertical pneumatic membrane peeler-cutter scissors (Figure 2).
Managing Potential Problems During Surgery
• Bleeding. Even with thorough preoperative planning, intraoperative bleeding can be cumbersome. Make sure to meticulously stop all bleeding foci as early as possible with gentle endodiathermy, endolaser and elevating intraocular pressure as needed.
• Avoiding and managing iatrogenic breaks. In nonrhegmatogenous diabetic TRDs, avoiding iatrogenic breaks is crucial, because these require a more aggressive approach and carry a worse long-term prognosis, including the possible development of proliferative vitreoretinopathy (PVR). If breaks occur, it’s imperative to relieve all surrounding traction and membranes, or make a focal retinectomy of surrounding inseparable plaques when peeling is not possible. In a few cases with large FVM plaques around breaks combined with PVR and vitreous base contraction, large (90 to 180 degrees) retinectomies and scleral buckle placement may be needed to release traction successfully. Demarcate all breaks with gentle endodiathermy to
ensure they are found and lasered accordingly after air-fluid
exchange.
• Air-fluid exchange around breaks. After completing membrane dissection and ruling out any residual hyaloid, perform an air-fluid exchange followed by PRP and laser retinopexy around breaks. It’s much easier to laser the peripheral anterior retina in the operating room as opposed to the clinic, so this is a great time to ensure good
peripheral laser is completed all the way to the ora serrata. We like to use flexible, curved, endoilluminated laser probes because they allow us to deliver anterior
PRP while performing scleral
depression.
• Gas with face-down positioning. At the end of the case, whether we encounter breaks or not, we like to use long-acting 14% or 16% perfluoropropane (C3F8) gas tamponade with prolonged face-down positioning of two to three weeks duration. We reserve silicone oil for very rare cases that need an extensive and large inferior retinectomy (about 5 percent of cases). This gas tamponade/face-down positioning approach has provided excellent results in one study of 89 consecutive, complex diabetic TRD cases amongst our team:13 an approximately 98-percent primary reattachment rate with a single surgery, and less than 2 percent secondary retinal detachments in complex cases.
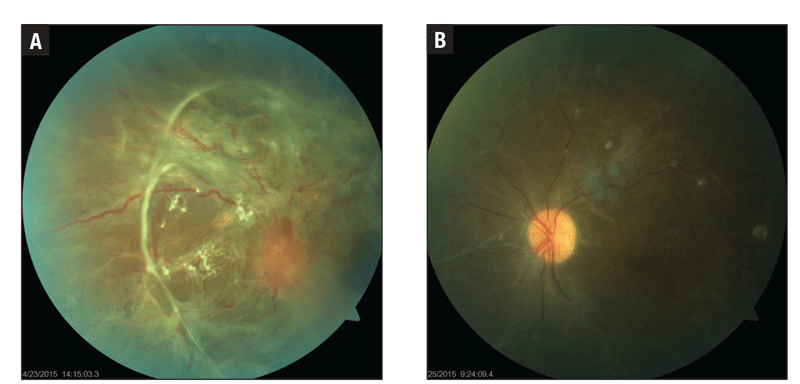 |
Figure 3. Preoperative visual acuity in this patient was counting fingers (A), but tractional retinal detachment repair restored VA to 20/80 (B). |
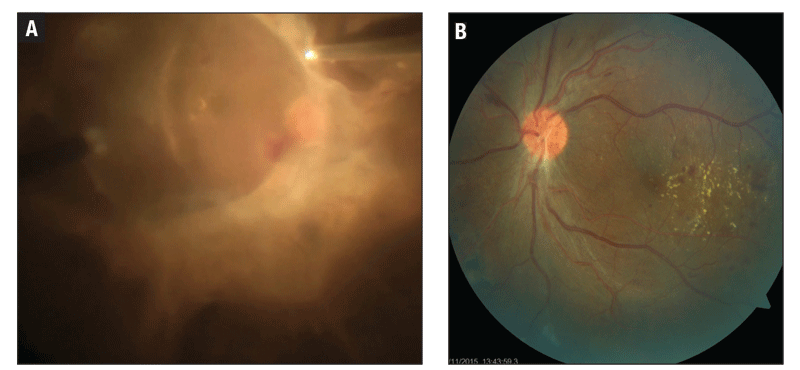 |
Figure 4. In this patient, preoperative visual acuity was counting fingers (A). Postoperative VA was 20/40 after tractional detachment repair (B). |
Gas with face-down positioning provides tamponade of possible undetected iatrogenic or laser-induced microbreaks while PRP scars are forming. Furthermore, in one study gas tamponade eliminated postoperative vitreous hemorrhage (VH) vs. a 17-percent VH rate in cases without it.14
Postoperative Care
• Role of postoperative steroids. At the conclusion of the case we frequently treat the patient with steroids to prevent postoperative fibrin and the inflammatory cascade, especially in cases with significant intraoperative PRP, membrane peeling and a long case duration. In addition to sub-Tenon’s triamcinolone, we ask anesthesia to give 125 mg of intravenous solumedrol if the patient’s blood-sugar levels are controlled, followed sometimes by an oral dose of prednisone in the postanesthesia care unit.
• Monitor for sequelae. We follow these patients very closely postoperatively, monitoring for possible redetachment, recurrent VH, fibrinoid syndrome and anterior hyaloid proliferation, neovascular glaucoma and progressing cataract. Recurrent VH following diabetic TRD repair is a common complication occurring in 16 to 43 percent of cases, with approximately 5 to 10 percent requiring a vitrectomy washout.15,16
• Address patient expectations. Lastly, the follow-up visits are important opportunities to maintain the patient’s expectations, because visual-acuity results can be highly variable.
Extra Effort is Priceless
Repairing diabetic TRDs is extremely complex and time-consuming, but this carefully planned and executed surgical approach can result in excellent outcomes (Figures 3 and 4). While these cases all have the same underlying disease process, each TRD and patient is unique. A personalized care plan is necessary. Improvements in the evolution of small-gauge vitreoretinal instrumentation, along with a continually growing base of knowledge and novel techniques, keep improving the success rates of these difficult surgeries.
As we’ve described, managing and treating diabetic TRDs is a long-term process encompassing the preoperative, intraoperative and postoperative periods, but the extra effort is priceless for providing good vision and hope for these sick diabetic patients.
REFERENCES
1. Charles S, Flinn CE. The natural history of diabetic extramacular tractional retinal detachment. Arch Ophthalmol. 1981;99:66–8.
2. Eliott D, Hemeida T. Diabetic traction retinal detachment. Int Ophthalmol Clin. 2009;49:153-165.
3. Thompson JT, de Bustros S, Michels RG, Rice TA. Results and prognostic factors in vitrectomy for diabetic traction retinal detachment of the macula. Arch Ophthalmol. 1987;105:497-502.
4. Thompson JT, de Bustros S, Michels RG, Rice TA. Results and prognostic factors in vitrectomy for diabetic traction-rhegmatogenous retinal detachment. Arch Ophthalmol. 1987;105:503-507.
5. Hwang JC, Sharma AG, Elliott D. Fellow eye vitrectomy for proliferative diabetic retinopathy in an inner city population. Br J Ophthalmol. 2013;97:297–301.
6. Shukla SY, Hariprasad AS, Hariprasad SM. Long-term mortality in diabetic patients with tractional retinal detachments. Ophthalmol Retina. 2017;1:8-11.
7. Zhao XY, Xia S, Chen YX. Antivascular endothelial growth factor agents pretreatment before vitrectomy for complicated proliferative diabetic retinopathy: a meta-analysis of randomised controlled trials. Br J Ophthalmol. 2018;102:1077-1085.
8. Rizzo S, Genovesi-Ebert F, Di Bartolo E, et al. Injection of intravitreal bevacizumab (Avastin) as a preoperative adjunct before vitrectomy surgery in the treatment of severe proliferative diabetic retinopathy (PDR). Graefes Arch Clin Exp Ophthalmol. 2008;246:837–842.
9. Modarres M, Nazari H, Falavarjani KG, et al. Intravitreal injection of bevacizumab before vitrectomy for proliferative diabetic retinopathy. Eur J Ophthalmol. 2009;19:848–852.
10. Khan MA, Samara WA, Hsu J, Garg S. Short-term outcomes of hybrid 23-, 25-, and 27-gauge vitrectomy for complex diabetic tractional retinal detachment repair. Retin Cases Brief Rep. 2017 Mar 29. [Epub ahead of print]
11. Gunduz K, Bakri SJ. Management of proliferative diabetic retinopathy. Compr Ophthalmol Update. 2007;8:245–256.
12. Berrocal MH. All-probe vitrectomy dissection techniques for diabetic tractional retinal detachments: Lift and Shave. Retina. 2018;38:S2-S4.
13. Dawood S, Georgiou M, Patel K, Skondra D. Outcomes of diabetic tractional retinal detachment repair In Chicago Cook County Hospital Health System: Initial experience of a new service. Invest Ophthalmol Vis Sci. 2016;57;6320.
14. Yang CM, Yeh PT, Yang CH. Intravitreal long-acting gas in the prevention of early postoperative vitreous hemorrhage in diabetic vitrectomy. Ophthalmology. 2007;114:710–715.
15. Lee DY, Jeong HS, Sohn HJ, et al. Combined 23-gauge sutureless vitrectomy and clear corneal phacoemulsification in patients with proliferative diabetic retinopathy. Retina. 2011;31:1753–1758.
16. Zhao LQ, Zhu H, Zhao PQ, et al. A systematic review and metaanalysis of clinical outcomes of vitrectomy with or without intravitreal bevacizumab pretreatment for severe diabetic retinopathy. Br J Ophthalmol. 2011;95:1216–22.



