 |
Viral retinitis is rare—0.63 cases per 1 million population per year—with equal incidence among males and females and a large variation in age of presentation.5,6 However, mixed-mechanism retinal detachment secondary to viral retinitis is common, occurring in 20 to 75 percent of these eyes, with resultant poor visual outcomes.7-10 These complex RDs require surgery. Here, we discuss our own study of surgical and anatomic outcomes in patients with RD secondary to viral retinitis11 and highlight a novel surgical technique called the “PFO-foscarnet pocket.”12
Etiology of Viral Retinitis
Diagnosis of viral retinitis is based on the American Uveitis Society clinical criteria that include:
Focal, well-demarcated areas of retinal necrosis in the peripheral retina.
Rapid and circumferential progression of necrosis.
Evidence of occlusive vasculopathy.
Prominent inflammatory reaction in the vitreous and anterior chamber.13
Etiology in immunocompetent patients is varicella zoster virus in 50 percent, herpes simplex virus in 25 percent, Epstein-Barr virus in 15 percent and cytomegalovirus (CMV) in 1 percent, although the latter is much more common in immunocompromised patients.14–16
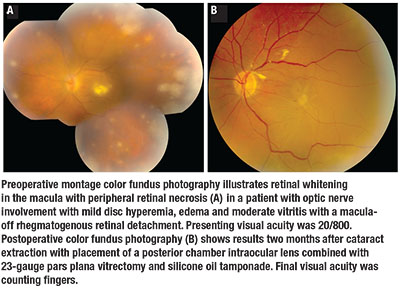 |
Oral agents like valacyclovir and famciclovir have superior bioavailability and central nervous system penetration compared to acyclovir and seem to have utility in both the initial treatment and maintenance therapy of patients with viral retinitis.3 A six-to-eight-week induction period followed by a three-to-six-month maintenance phase seems to be effective in this often chronic and recurrent clinical course.
 |
RDs in Viral Retinitis
RDs in viral retinitis usually occur after the acute phase of infection and are secondary to vitreous traction on the necrotic retina and inflammatory membranes, resulting in stretch holes and a partially rhegmatogenous component.27
In our study, all cases underwent three-port pars plana vitrectomy (PPV) with induction of a posterior vitreous detachment, if not already present. We employed membrane peeling, retinotomies and retinectomies to remove inflammatory vitreal and retinal membranes as well as necrotic retina (video available at bit.ly/2eKmGzW), and performed tamponade with 1,000-centistoke silicone oil in 11 of 12 eyes. During PPV, we placed a scleral buckle in 10 eyes (83.3 percent).
Initial surgery achieved successful anatomic re-attachment of the retina in all cases, and no re-detachments occurred during the follow-up period. In 50 percent of eyes, we successfully removed silicone oil at a later date with no recurrence of retinal detachment during the study period.
Foscarnet During Surgery
| To view a video that demonstrates the technique Drs. Almeida and Chin discuss, click here. |
Unlike acyclovir or gancyclovir, antiviral foscarnet does not require thymidine kinase activation, so resistance is uncommon. Foscarnet selectively inhibits viral polymerase and can rapidly inactivate the virus with a short contact exposure time. Consequently, we have successfully incorporated this therapy at the time of surgical repair.12 The steps in the technique are:
• Multiplane chromovitrectomy. This step facilitates retinal detachment repair with concurrent intraoperative intravitreal antiviral therapy prior to silicone oil tamponade.
• After removal of all vitreous and necrotic retina, injection of perfluoro-n-octane (PFO) liquid over the posterior pole. This flattens any posteriorly detached retina up to the most posterior edge of any breaks, creating a PFO-infusion fluid meniscus. At this point, there are two distinct phases in the vitreous cavity: a PFO bubble over the posterior pole; and a layer of balanced salt solution (BSS) more anteriorly. The PFO flattens and immobilizes the retina more posteriorly in its usual manner.
• Injection of approximately 0.1 to 0.2 mL of foscarnet (2.4 mg per 0.1 mL) over the PFO into the BSS more anteriorly. This creates a PFO-foscarnet “pocket” or “shell” that allows the foscarnet to penetrate the retina while the PFO stabilizes the posterior retina.
• At this point, laser retinopexy or any additional vitrectomy or retinectomy can be performed. If needed, this will allow the intravitreal foscarnet to have increased exposure time to the diseased peripheral retina.
• Air-fluid exchange. Upon completion of the vitrectomy, a soft-tipped extrusion cannula helps to perform an air-fluid exchange by first removing the anterior BSS-foscarnet followed by the more posterior PFO.
• Foscarnet wash. In the final step, injection of three to five drops of foscarnet over the posterior pole in the air-filled eye performs a foscarnet wash. This allows the foscarnet to penetrate any posterior retina previously inaccessible owing to the PFO placement. The silicone-tip extrusion needle helps to remove the foscarnet and any remaining BSS. The technique allows stabilization of the detached retina while allowing for antiviral washout during vitrectomy.
 |
We identified optic nerve involvement as a poor prognostic factor because only eyes without optic nerve involvement were able to achieve visual acuity of 20/100 or better. We used the proposed absolute criteria for optic nerve involvement in ARN to determine optic nerve involvement according to three criteria:
Afferent papillary defect not consistent with the retinal findings.
Poor correlation between retinal findings and visual acuity.
Sudden deterioration of visual acuity to 20/100 or worse without corresponding retinal changes within a 24- to 36-hour interval.28
Visual acuity seems unlikely to improve in these cases. Subsequent interventions primarily aim to prevent involvement of the fellow eye, chronic retinal detachment and phthisis bulbi.
Our novel PFO-foscarnet pocket allows for intraoperative antiviral coverage in this challenging group of patients, who often require long-acting silicone oil tamponade. RS
REFERENCES
1. Urayama A, Yamada N, Sasaki T. Unilateral acute uveitis with retinal periarteritis and detachment [In Japanese]. Rinsho ganka. 1971;25:607-619.
2. Willerson D, Jr., Aaberg TM, Reeser FH. Necrotizing vaso-occlusive retinitis. Am J Ophthalmol. 1977;84:209-219.
3. Tibbetts MD, Shah CP, Young LH, et al. Treatment of acute retinal necrosis. Ophthalmology. 2010;117:818-824.
4. Young NJ, Bird AC. Bilateral acute retinal necrosis. Br J Ophthalmol. 1978;62:581-590.
5. Cochrane TF, Silvestri G, McDowell C, et al. Acute retinal necrosis in the United Kingdom: results of a prospective surveillance study. Eye (Lond). 2012;26:370-377; quiz 378.
6. Hillenkamp J, Nölle B, Bruns C, et al. Acute retinal necrosis: clinical features, early vitrectomy, and outcomes. Ophthalmology. 2009;116:1971-1975 e1972.
7. Sims JL, Yeoh J, Stawell RJ. Acute retinal necrosis: a case series with clinical features and treatment outcomes. Clin Experiment Ophthalmol. 2009;37:473-477.
8. Meghpara B, Sulkowski G, Kesen MR, et al. Long-term follow-up of acute retinal necrosis. Retina. 2010;30:795-800.
9. Tran TH, Stanescu D, Caspers-Velu L, et al. Clinical characteristics of acute HSV-2 retinal necrosis. Am J Ophthalmol. 2004;137:872-879.
10. Lau CH, Missotten T, Salzmann J, Lightman SL. Acute retinal necrosis features, management, and outcomes. Ophthalmology. 2007;114:756-762.
11. Almeida DRP, Chin EK, Tarantola RM, et al. Long-term outcomes in patients undergoing vitrectomy for retinal detachment due to viral retinitis. Clin Ophthalmol. 2015;9:1307-14.
12. Yu K, Chin EK, Mahajan VB, Almeida DRP. Intravitreal foscarnet with concurrent silicone oil tamponade for rhegmatogenous retinal detachment secondary to viral retinitis. Retina. Accepted for publication May 2016.
13. Holland GN, Executive Committee of the American Uveitis Society. Standard diagnostic criteria for the acute retinal necrosis syndrome. Am J Ophthalmol. 1994;117:663-667.
14. Ganatra JB, Chandler D, Santos C, et al. Viral causes of the acute retinal necrosis syndrome. Am J Ophthalmol. 2000;129:166-172.
15. Walters G, James TE. Viral causes of the acute retinal necrosis syndrome. Curr Opin Ophthalmol. 2001;12:191-195.
16. Kanoff J, Sobrin L. New diagnosis and treatment paradigms in acute retinal necrosis. Int Ophthalmol Clin. 2011;51:25-31.
17. Tran TH, Rozenberg F, Cassoux N, et al. Polymerase chain reaction analysis of aqueous humour samples in necrotising retinitis. Br J Ophthalmol. 2003;87:79-83.
18. Gargiulo F, De Francesco MA, Nascimbeni G, et al. Polymerase chain reaction as a rapid diagnostic tool for therapy of acute retinal necrosis syndrome. J Med Virol. 2003;69:397-400.
19. Sugita S, Shimizu N, Watanabe K, et al. Use of multiplex PCR and real-time PCR to detect human herpes virus genome in ocular fluids of patients with uveitis. Br J Ophthalmol. 2008;92:928-932.
20. Asano S, Yoshikawa T, Kimura H, et al. Monitoring herpes virus DNA in three cases of acute retinal necrosis by real-time PCR. J Clin Virol. 2004;29:206-209.
21. Cottet L, Kaiser L, Hirsch HH, Baglivo E. HSV2 acute retinal necrosis: diagnosis and monitoring with quantitative polymerase chain reaction. Int Ophthalmol 2009;29:199-201.
22. Emerson GG, Smith JR, Wilson DJ, et al. Primary treatment of acute retinal necrosis with oral antiviral therapy. Ophthalmology. 2006;113:2259-2261.
23. Blumenkranz MS, Culbertson WW, Clarkson JG, Dix R. Treatment of the acute retinal necrosis syndrome with intravenous acyclovir. Ophthalmology. 1986;93:296-300.
24. Aslanides IM, De Souza S, Wong DT, et al. Oral valaciclovir in the treatment of acute retinal necrosis syndrome. Retina. 2002;22:352-354.
25. Chong DY, Johnson MW, Huynh TH, et al. Vitreous penetration of orally administered famciclovir. Am J Ophthalmol. 2009;148:38-42 e31.
26. Palay DA, Sternberg P, Jr., Davis J, et al. Decrease in the risk of bilateral acute retinal necrosis by acyclovir therapy. Am J Ophthalmol. 1991;112:250-255.
27. McDonald HR, Lewis H, Kreiger AE, et al. Surgical management of retinal detachment associated with the acute retinal necrosis syndrome. Br J Ophthalmol. 1991;75:455-458.
28. Sergott RC, Anand R, Belmont JB, et al. Acute retinal necrosis neuropathy. Clinical profile and surgical therapy. Arch Ophthalmol. 1989;107:692-696.



