The advent of panretinal photocoagulation has helped to significantly reduce the risk of vision loss secondary to proliferative diabetic retinopathy.1 Recent trials have shown that intravitreal anti-VEGF therapy has a comparable, if not superior, treatment effect on PDR compared to PRP.2,3 However, both therapies require consistent post-treatment follow-up.
 |
Although studies have evaluated adherence to dilated fundus examination regimens established for diabetic patients,4 limited data exists on loss to follow-up (LTFU) after treatment among these patients. Therefore, we conducted a study, recently published in the journal Ophthalmology,5 that sought to evaluate LTFU after either PRP or anti-VEGF therapy for PDR and identify key independent predictors of LTFU. Here, we report on what the study revealed about patients who don’t return for follow-up after treatment.
Study Findings
Our final analysis included 2,302 patients, of whom 1,272 (55.3 percent) received PRP and 1,030 (44.7 percent) received anti-VEGF treatment. The mean (standard deviation [SD]) number of PRP sessions was two (±1.3), and the mean number of intravitreal anti-VEGF injections given was 3.8 (±4.5). There were 584 (25.4 percent) patients LTFU immediately post-treatment and 1,718 (74.6 percent) that followed up within 12 months.
 |
Risk Factors of LTFU
We observed several risk factors that were significantly associated with LTFU.
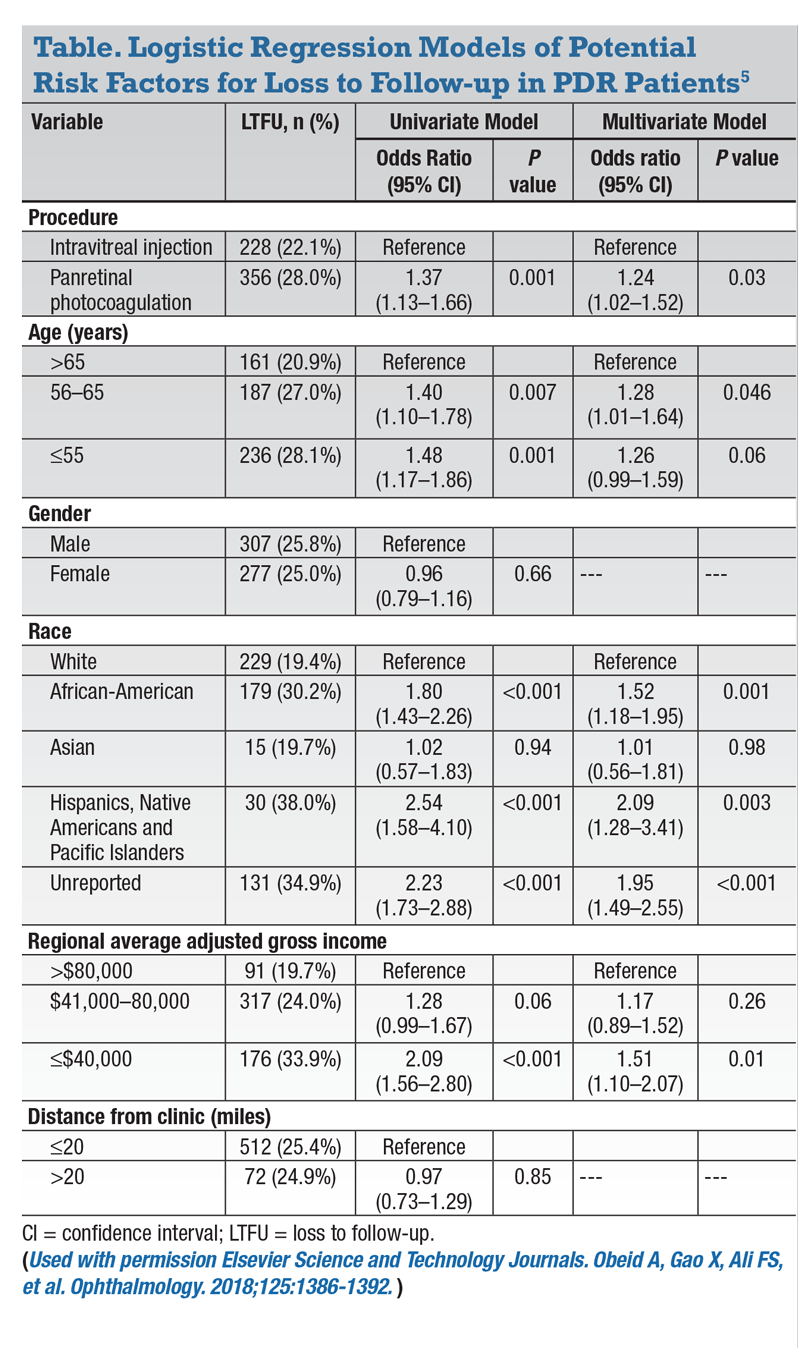
• Type of procedure. The first risk factor observed was the type of procedure, with 356 patients (28 percent) who received PRP and 228 (22.1 percent) who received anti-VEGF therapy lost to follow-up post treatment (p=0.001).
One can postulate the higher rate observed in the PRP group is secondary to selection bias, because physicians are more likely to attempt to select patients who they think would be compliant for anti-VEGF therapy. This appears to partially account for the disparity, as the effect of the procedure on LTFU odds
declines after adjusting for the various socio-demographic factors in the multivariate model (Table).
 |
However, LTFU may also be secondary to other factors, such as pain level differences that patients experience between the two treatments. One study has shown that patients receiving PRP experience pain levels approximately 60 times higher on average than intravitreal injections when quantified on a visual analog scale.6 Moreover, complications, such as diabetic macular edema and vitreous hemorrhage, which are more often associated with PRP, may also discourage patients from returning for follow-up.2,3
 |
• Age. Rates of LTFU also decreased as age increased, with rates of 28.1 percent, 27 percent and 20.9 percent for patients ages ≤55, 56 to 65 and >65 years old, respectively (p=0.002). Interestingly, it appears the major decline in LTFU rates occurs after the age of 65.
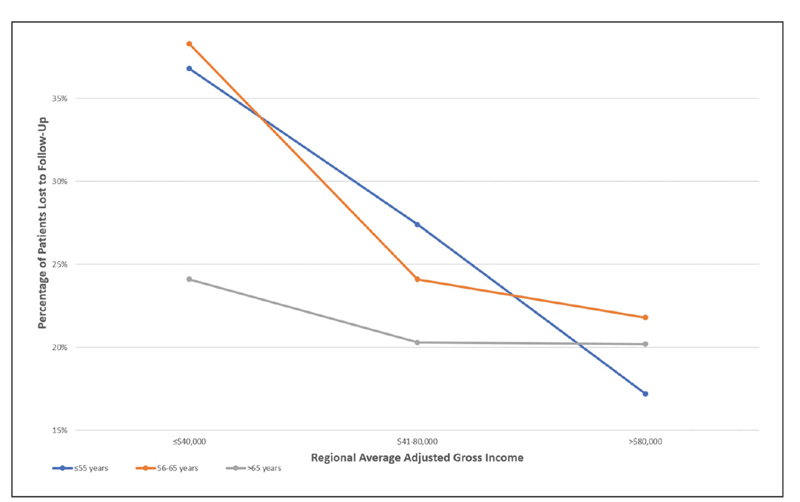 |
| Figure. Change in loss to follow-up (LTFU) rates by age over approximately four years stratified for regional average adjusted gross income. (Used with permission Elsevier Science and Technology Journals. Obeid A, Gao X, Ali FS, et al. Ophthalmology. 2018;125:1386-1392.) |
 |
We theorize that this may be related to insurance coverage as individuals age 65 and older are Medicare eligible. If this is true, we would expect patients of lower incomes to have a more pronounced disparity in LTFU rates when compared to higher incomes for patients younger than 65. Indeed, this is what the study observed, with significant differences in LTFU rates between age groups for regional adjusted gross incomes of ≤$40,000 (p=0.02) and $41,000 to $80,000 (p=0.04), but not for incomes of over $80,000 (p=0.62) (Figure). We also hypothesized that this may be due to the amount of time patients have available to see doctors; older individuals are much less likely to have a full-time job.
• Race. African-Americans had significantly higher LTFU rates (30.2 percent) than whites (19.4 percent) and Asians (19.7 percent) (p <0.001).
The evidence regarding the association of race and compliance with follow-up remains inconclusive, with conflicting findings from different studies.4,7,8
Underlying etiologies that contribute to the differences in LTFU rates may include distrust in the health-care system,9 access to insurance coverage and time.10 However, the number of social and environmental factors involved may render such an evaluation difficult.
Interestingly, we also observed an increase in LTFU rates in patients who refused to identify their race (34.9 percent). We believe this may represent a psychosocial component, and these patients may be more skeptical of the health-care system. We need further studies to understand this cohort and the true etiology contributing to high LTFU rates.
Finally, all significant risk factors in the univariate model were retained in the multivariate model as significant independent predictors of LTFU (Table, page 41).
Role of Visual Acuity
Contrary to our expectations, VA did not appear to play a significant role in LTFU rates. LTFU rates for eyes with ≥20/40, 20/50 to 20/200, and <20/200 VA at the final procedure before LTFU were 15 percent, 18.8 percent and 16.5 percent, respectively (p=0.37, n=920).
Given that our analysis only included a subgroup of patients with accessible medical records, the lack of a difference may be secondary to sample size. However, other studies also appear to present conflicting findings on the role of VA in LTFU,11,12 although these studies evaluated different diseases and used different definitions of LTFU.
Moreover, there did not appear to be a difference in baseline VA by follow-up status in the Diabetic Retinopathy Clinical Research Network Protocol S five-year follow-up results.13 Therefore, we will need further studies to validate the true role of VA on LTFU rates.
Factors That Hinder Follow-up
Our study results demonstrated that more than 20 percent of patients with PDR were LTFU after at least one treatment session over a four-year period. Although limited evidence exists on LTFU rates in patients with PDR, previous studies have documented high rates of noncompliance with recommended guidelines in diabetic patients. For example, although patients with diabetes require at least one dilated fundus exam annually (as the practice guidelines recommend),14 more than a fourth of patients have a year or less with a documented dilated fundus exam over four consecutive years of follow-up.7
Patients with DME have also shown high noncompliance with follow-up rates in Europe.15 More recently, the five-year results of the Protocol S trial showed that approximately 40 percent of patients were LTFU from both groups over the duration of the trial.15 This is particularly concerning, as we would expect that patients who agree to take part in such trials are generally more concerned with their disease and more likely to be motivated to comply with follow-up recommendations.
LTFU rates become even more relevant when we consider the potential sequelae secondary to LTFU stratified by treatment selection. Another study we recently published reported that eyes LTFU post-intravitreal anti-VEGF therapy fare much worse, both anatomically and functionally, when compared to eyes that received PRP.16
A final important point to consider is the limited life expectancy of patients with PDR. Studies have shown high mortality rates in the patients that received PRP in the original Early Treatment Diabetic Retinopathy Study.17 Moreover, these patients are expected to have numerous additional medical check-ups for other pathology secondary to the diabetes. Given the durability differences between the two treatments, with PRP demonstrated to have long-lasting effects, it becomes important to carefully evaluate each patient before selecting treatment.
 |
Future Directions
Although our study has identified several risk factors associated with LTFU, much remains to be done. Models evaluating social behavior are less robust than models evaluating physiological factors, given the numerous factors and complex confounders involved.
Although we assume we understand the factors most important in bringing back patients to our clinics, our current data demonstrates that this might not necessarily be true. This becomes particularly relevant in designing screening protocols that identify high-risk patients and interventions that aim to reduce LTFU rates. Models that have good predictive potential can help in achieving both these goals and provide a much better understanding on what contributes most to LTFU in our population.
REFERENCES
1. No authors listed. Photocoagulation treatment of proliferative diabetic retinopathy. Clinical application of Diabetic Retinopathy Study (DRS) findings, DRS Report Number 8. The Diabetic Retinopathy Study Research Group. Ophthalmology 1981;88:583–600.
2. Sivaprasad S, Prevost AT, Vasconcelos JC, et al for the Clarity Study Group. Clinical efficacy of intravitreal aflibercept versus panretinal photocoagulation for best corrected visual acuity in patients with proliferative diabetic retinopathy at 52 weeks (CLARITY): a multicentre, single-blinded, randomised, controlled, phase 2b, non-inferiority trial. Lancet Lond Engl. 2017;389:2192-2203.
3. Gross JG, Glassman AR, Jampol LM, et al. Panretinal photocoagulation vs intravitreous ranibizumab for proliferative diabetic retinopathy: A randomized clinical trial. JAMA. 2015;314:2137–2146.
4. Sloan FA, Yashkin AP, Chen Y. Gaps in receipt of regular eye examinations among medicare beneficiaries diagnosed with diabetes or chronic eye diseases. Ophthalmology. 2014;121:2452–2460.
5. Obeid A, Gao X, Ali FS, Talcott KE, Aderman CM, Hyman L, Ho AC, Hsu J. Loss to follow-up in patients with proliferative diabetic retinopathy after panretinal photocoagulation or intravitreal anti-VEGF injections. Ophthalmology. 2018;125:1386-1392.
6. Lucena CRF de A, Ramos Filho JA, Messias AMV, et al. Panretinal photocoagulation versus intravitreal injection retreatment pain in high-risk proliferative diabetic retinopathy. Arq Bras Oftalmol. 2013;76:18–20.
7. Sloan FA, Brown DS, Carlisle ES, et al. Monitoring visual status: why patients do or do not comply with practice guidelines. Health Serv Res. 2004;39:1429–1448.
8. Jiang X, Varma R, Torres M, et al. Self-reported use of eye care among adult Chinese Americans: The Chinese American Eye Study. Am J Ophthalmol. 2017;176:183–193.
9. Armstrong K, Ravenell KL, McMurphy S, Putt M. Racial/ethnic differences in physician distrust in the United States. Am J Public Health. 2007;97:1283–1289.
10. Ellish NJ, Royak-Schaler R, Passmore SR, Higginbotham EJ. Knowledge, attitudes, and beliefs about dilated eye examinations among African-Americans. Invest Ophthalmol Vis Sci. 2007;48:1989–1994.
11. Saadine JB, Fong DS, Yao J. Factors associated with follow-up eye examinations among persons with diabetes. Retina. 2008;28:195–200.
12. Droege KM, Muether PS, Hermann MM, et al. Adherence to ranibizumab treatment for neovascular age-related macular degeneration in real life. Graefes Arch Clin Exp Ophthalmol. 2013;251:1281–1284.
13. Gross JG, Glassman AR, Liu D, et al. Five-year outcomes of panretinal photocoagulation vs intravitreous ranibizumab for proliferative diabetic retinopathy: A randomized clinical trial. JAMA Ophthalmol. 2018 July 24 [epub ahead of print].
14. Diabetic Retinopathy PPP (Preferred Practice Pattern) - Updated 2017. American Academy Ophthalmology. Available at: https://www.aao.org/preferred-practice-pattern/diabetic-retinopathy-ppp-updated-2017. Accessed September 27, 2018.
15. Weiss M, Sim DA, Herold T, et al. Compliance and adherence of patients with diabetic macular edema to intravitreal anti-vascular endothelial growth factor therapy in daily practice. Retina. 2017 October 24[epub ahead of print].
16. Obeid A, Su D, Patel SN, et al. Outcomes of eyes lost to follow-up with proliferative diabetic retinopathy that received panretinal photocoagulation vs intravitreal anti-vascular endothelial growth factor. Ophthalmology. Published online August 2, 2018 [Epub ahead of print].
17. Chew EY, Ferris FL, Csaky KG, et al. The long-term effects of laser photocoagulation treatment in patients with diabetic retinopathy: the early treatment diabetic retinopathy follow-up study. Ophthalmology. 2003;110:1683–1689.



