 |
Three-dimensional, digitally assisted visualization systems are enhancing optical microscope-based approaches to vitreoretinal surgery. Besides the clear advantages of the 3-D technology over the traditional approach—4,000-pixel (4K) monitor, decreased light phototoxicity, digital enhancements, improved depth of field, digital filtering and high-dynamic range—these platforms can be integrated with other commercially available visualization systems.1,2
Available 3-D systems include:
- Ngenuity 3-D Visualization System (TrueVision Systems and Alcon);
- Trenion 3-D HD (Carl Zeiss Meditec); and
- RV800 Viewing System (Leica Microsystems).
Currently, Ngenuity offers all the described features, including integration of endoscopy and intraoperative optical coherence tomography.
At a debate during the American Academy of Ophthalmology’s Retina Subspecialty Day last year, the audience voted on whether 3-D digitally assisted vitreoretinal surgery is ready to become the new standard. Although the audience voted largely no, the arguments were based only on image quality and teaching advantages. The digital integration with other technologies, though, is something unprecedented in our field, especially with regards to endoscopic vitrectomy. 3,4 Here, we report on the state of the art of 3-D digitally assisted platforms in vitreoretinal surgery.
 |
Evolution of Endoscopy
Harvey Thorpe, MD, first described endoscopic ocular surgery in 1934,5 well before pars plana vitrectomy became the gold standard in vitreoretinal surgery. In the early 1990s, Martin Uram, MD, at New York Eye and Ear Infirmary, introduced endoscopy to the vitreoretinal world.6 Despite slow initial progress in the developing this technology, it evolved toward the higher-resolution (17,000 pixels) and smaller-gauge probes (23-gauge in North America [Endo Optiks E2 endoscope platform, Beaver-Visitec International Inc.] and 25-ga in Japan [FiberTech Co. Ltd.]). This advance in technology made endoscopy more attractive to vitreoretinal surgeons.
However, endoscopic vitrectomy is still only being adopted by a few retina specialists. The learning curve can be steep, and a misconception exists that endoscopic vitrectomy has only a limited number of indications.
Advantages of Endoscopy
The advantages of endoscopy in cases of media opacity, such as cloudy cornea, trauma or endophthalmitis are well known, and it’s accepted as an alternative to temporary keratoprosthesis or “blind vitrectomy.
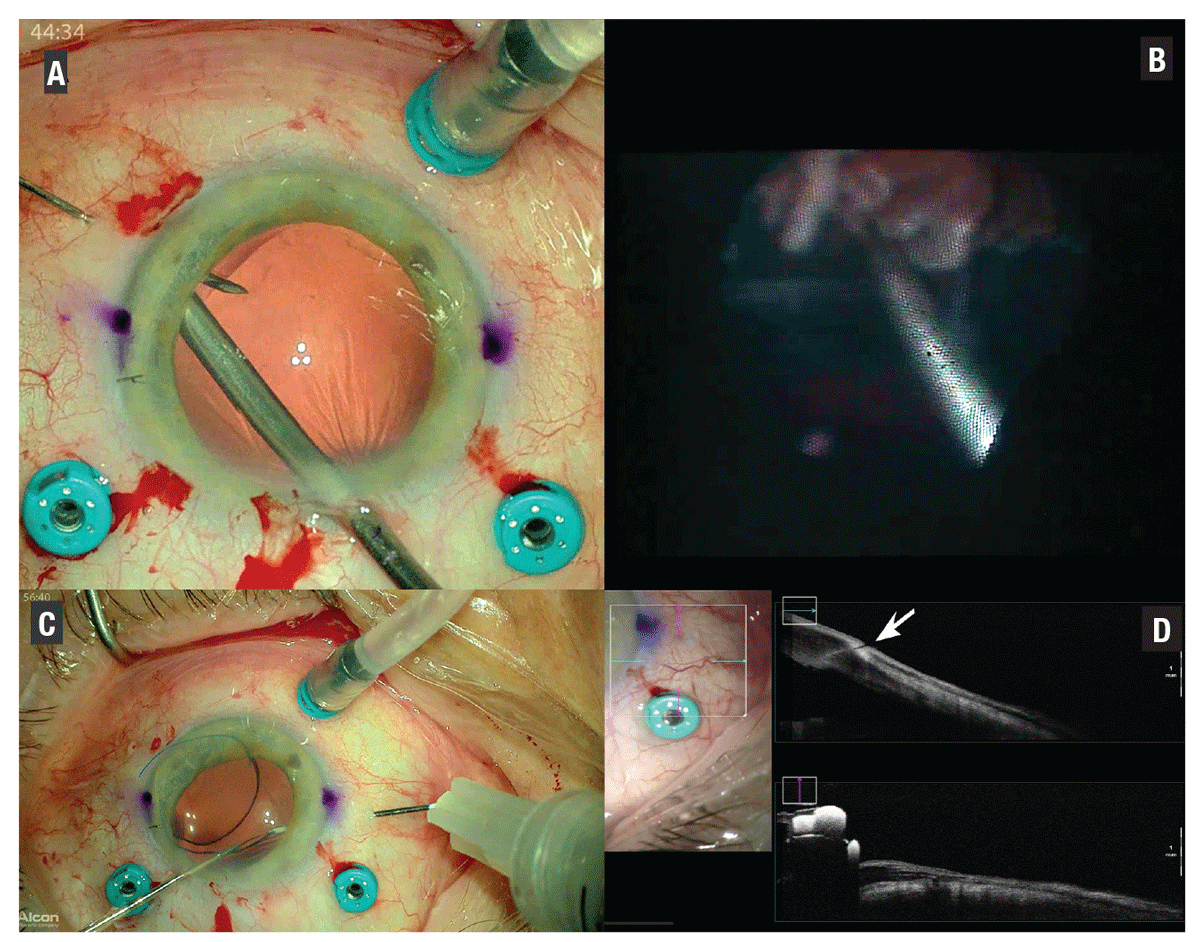 |
| Figure 1. Images show secondary intraocular lens implantation with sutureless intrascleral IOL fixation. The split screen from the Ngenuity 3D Visualization System combines wide-field visualization (A) and endoscopic view (B) using a 19-gauge endoscope through the corneal wound in an aphakic eye. During needle insertion, the endoscope confirms the intraocular entrance posterior to the ciliary processes. The first haptic is externalized with the 27-ga needle (C). Intraoperative optical coherence tomography (D) confirms the position of the intrascleral haptic (arrow). |
Surgeons unfamiliar with endoscopy may be unaware of the advantages of visualizing structures between the ora serrata and the retro-iridial space. Scleral depression or other technologies, such as the Topcon OMS-800 OFFISS microscope visualization system available in some countries, allow us to access the far periphery of the retina up to the ora serrata for shaving the vitreous base or peeling membranes. But they poorly visualize more anterior anatomical structures, which could aid in recognizing and diagnosing underlying pathology. We don’t know what we don’t know.
The availability of small-gauge endoscopy probes represented a big step forward. One of the advantages of a small-gauge endoscope is that it allows the surgeon to switch hands and reposition from superior to temporal trocars when he/she needs to address pathology in different locations.
In addition, valved small-gauge trocars also help maintain the fluidics control in more complex cases, representing an advantage over the larger-gauge trocars (19- and 20-ga) that glaucoma specialists use for endocyclophotocoagulation. The indications in clear media further extend the indications when media opacity is present (Table).11-17
 |
Besides the limitations of straight small-gauge instruments when working in the far periphery, endoscopic visualization does pose some challenges for the newcomer. They include poor image resolution and limited field of view in a 2-D environment (lack of stereopsis), making it harder to find instruments inside the eye, which may discourage adoption. Consequently, few reports support its use. In addition, terminology used in both clinical scenarios (media opacity or clear media) should be reappraised.
Integration of Endoscopy, 3-D
The integration of endoscopy into 3-D visualization systems offers a new perspective to surgeons with little to no endoscopy experience, helping them to overcome the learning curve faster and allowing them to work simultaneously in a familiar wide-field environment.
The new 3-D monitors with split screen combine endoscopic and wide-field view simultaneously during endoscopic vitrectomy. Traditionally, the use of the endoscope requires a monitor. The surgeon works heads up, looking at the screen (2-D) instead of through the microscope. The 3-D, heads-up technology eliminates the need for a second monitor because the probe is connected to the 3-D system through a coaxial output from the endoscopy console to the 4K monitor.
 |
Alternatively, other devices are available to convert S-Video output from the endoscope into HDMI. The Ngenuity split-screen feature simplifies the surgical setup. The endoscopy probe simultaneously provides at the same time the endoscopic image and the wide-field 3-D viewing as an endoilluminator (Figure 2).
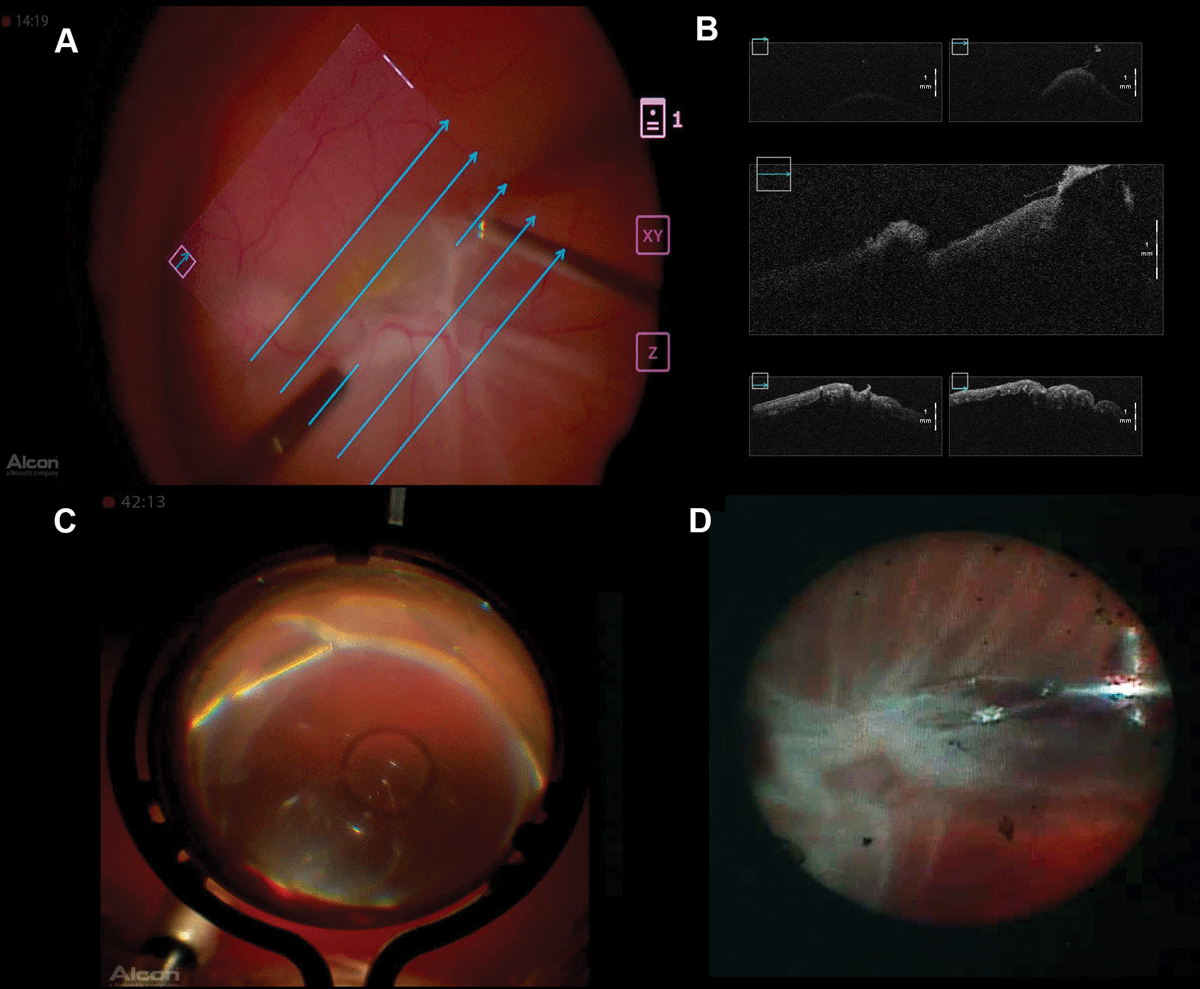 |
| Figure 2. Images show treatment of a recurrent retinal detachment with hypotony and proliferative vitreoretinopathy C-1 and C-5 that involves macular star fold peeling with intraocular forceps assisted with intraoperative optical coherence tomography (A). Intraoperative OCT (B) shows real-time PVR peeling. Ngenuity split screen combines wide-fi eld visualization (C) and endoscopic view (D) using a 23-gauge endoscopy probe and 25-ga adjustable chandelier to peel the anterior PVR causing traction and hypotony/ |
The most challenging aspects in learning endoscopic vitrectomy include having the right orientation, being aware of the distance from ocular tissues and positioning the
intraocular instruments. Traditionally during endoscopy, if the surgeon is struggling to find where the instruments are during the endoscopic view, one has to go back to the microscope to confirm the position or feel comfortable with the view. This takes a few seconds and annoys surgeons because of the need to readjust when they switch from the microscope to the endoscopy monitor and back.
However, the split-screen feature can allow the surgeon to skip this step because this information is readily available at any time during the surgery. Using the Ngenuity cursor, you can activate the split-screen feature, allowing simultaneous viewing of endoscopy and a wide-field 3-D image (Figure 3). In addition, new Ngenuity Datafusion software integrates the Constellation Vision System (Alcon), which allows surgeons to track key data parameters (intraocular pressure, flow rates, infusion pressure and laser power) in real-time, and offers additional functionality that allows for customization.
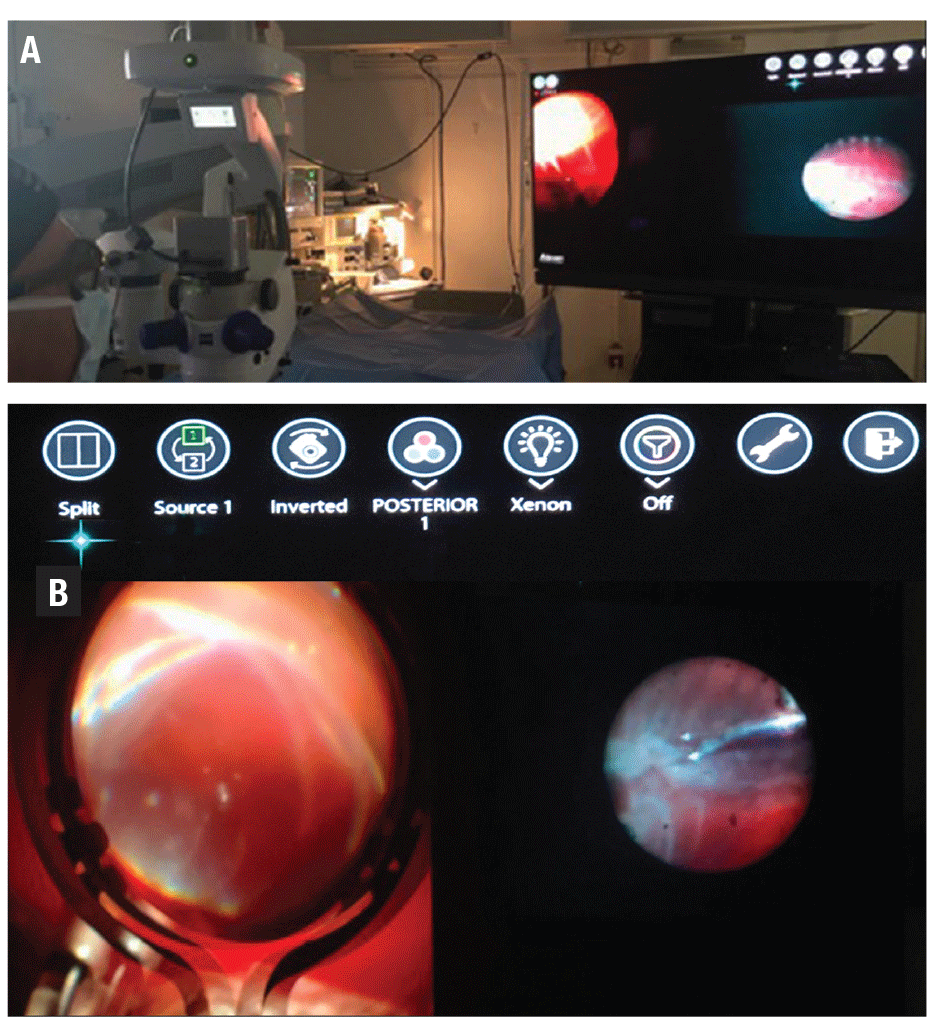 |
Figure 3. Operating-room setup of the Ngenuity 3D Visualization System (A)with |
Using 2-D viewing in a 4K, 3-D platform substantially improves image quality and allows a better detection of tissue planes despite the inherent limitations of fiber optics in a small-gauge probe, although the stereopsis doesn’t change. The lighting systems in endoscopy tend to cause glare, although digital image enhancement can reduce the glare and confer additional advantages to this system.
 |
Looking Ahead
The application of a 3-D digitally assisted imaging system to vitreoretinal surgery is one of the latest and most promising advances in our field. Combining improved image performance with technologies such as endoscopic vitreoretinal surgery, intraoperative OCT and video overlay features should help improve surgical precision and, ultimately, outcomes for our patients.
REFERENCES
1. Eckardt C, Paulo EB. Heads-up surgery for vitreoretinal procedures: an experimental and clinical study. Retina. 2016;36:137–147.
2. Adam MK, Thornton S, Regillo CD et al. Minimal endoillumination levels and display luminous emittance during three-dimensional heads-up vitreoretinal surgery. Retina. 2017;37:1746-1749.
3. Morishita S, Kita M, Yoshitake S et al. 23-gauge vitrectomy assisted by combined endoscopy and a wide-angle viewing system for retinal detachment with severe penetrating corneal injury: A case report. Clin Ophthalmol. 2011;5:1767–1770.
4. Kita M, Mori Y, Hama S. Hybrid wide-angle viewing-endoscopic vitrectomy using a 3-D visualization system. Clin Ophthalmol. 2018;12:313-317.
5. Thorpe H. Ocular endoscope: Instrument for removal of intravitreous non-magnetic foreign bodies. Trans Am Acad Ophthalmol Otolaryngol. 1934;39: 422-424.
6. Uram M. Ophthalmic laser microendoscope endophoto-coagulation. Ophthalmology 1992;99:1829-1832.
7. Sabti KA and Raizada S. Endoscope-assisted pars plana vitrectomy in severe ocular trauma. Br J Ophthalmol. 2012;96: 1399-1403.
8. Huang YF, Chang CJ. Endoscope-assisted vitrectomy in the management of retinal detachment with corneal opacity. Taiwan J Opthalmol. 2017;7:164-167.
9. Boscher C. Endoscopic vitreoretinal surgery of the injured eye. In: Alfaro DV, Liggett PE, eds. Vitreoretinal Surgery of the Injured Eye. Philadelphia, PA: Lippincott-Raven; 1999: 301-314.
10. De Smet MD, Carlborg EA. Managing severe endophthalmitis with the use of an endoscope. Retina. 2005; 25:976–980.
11. Sonoda Y, Yamakiri K, Sonoda S, et al. Endoscopy-guided subretinal fluid drainage in vitrectomy for retinal detachment. Ophthalmologica. 2006; 220:83–86.
12. Sasahara M, Kiryu J, Yoshimura N. Endoscope-assisted transscleral suture fixation to reduce the incidence of intraocular lens dislocation. J Cataract Refract Surg. 2005; 31:1777–1780.
13. Sabti KA, S. Raizada JA, Kandari V et al. Applications of endoscopy in vitreoretinal surgery. Retina. 2008; 28: 159-166.
14. Lee GD, Goldberg RA, Heier JS. Endoscopy-assisted vitrectomy and membrane dissection of anterior proliferative vitreoretinopathy for chronic hypotony after previous retinal detachment repair. Retina. 2016;36:1058-1063.
15. Hammer ME, Grizzard WS. Endoscopy for evaluation and treatment of the ciliary body in hypotony. Retina. 2003; 23:30–36.
16. Boscher C, Lebuisson DA, Lean JS, et al. Vitrectomy with endoscopy for management of retained lens fragments and/or posteriorly dislocated intraocular lens. Graefes Arch Clin Exp Ophthalmol. 1998;236:115-121.
17. Dirani A, Ciongoli MR, Lesk MR, et al. Small-gauge endoscopy-guided pneumatic anterior hyaloid detachment: A new surgical technique for combined pars plana vitrectomy and pars plana glaucoma drainage implant. Ophthalmic Surg Lasers Imaging Retina. 2018;49:48-50.



