 |
The
| Clinical Sites Participating in the Macular Telangiectasia (MacTel) Project Australia Center for Eye Research Australia Lions Eye Institute Save Sight Institute Israel The Goldschleger Eye Institute France Hopital Lariboisiere Switzerland Klinik und Poliklinik fur Augenheilkunde Germany Universitats-Augenklinik Bonn St. Franziskus Hospital United Kingdom Moorfields Eye Hospital United States California Scripps Research Institute Jules Stein Eye Institute, UCLA Florida Bascom Palmer Eye Institute, University of Miami Massachusetts Massachusetts Eye and Ear Infirmary Michigan University of Michigan, Kellogg Eye Center New York Manhattan Eye, Ear & Throat Hospital The New York Eye and Ear Infirmary Ohio Retina Associates of Cleveland, Inc. Pennsylvania Scheie Eye Institute Utah University of Utah Medical Center Virginia The Retina Group of Washington Wisconsin University of Wisconsin |
However, the visual prognosis after ERM removal may not be good, especially for patients who had been treated for macula-sparing RRDs, which are not expected to cause visual impairment when treated successfully. A previous study reported that mean Snellen visual acuity was 20/40 after subsequent vitrectomy to remove an ERM that developed after repair of macula-sparing RRD.8 We recently reported that final visual acuity of 20/20 or better was achieved in only three of seven cases after removal of symptomatic post-surgical ERMs secondary to macula-sparing RRD.2
Another possible consequence of removal of ERMs is persistent metamorphopsia. Taking these facts into consideration, prevention of post-surgical ERM is desirable as long as it is achieved via a safe and practical procedure.
Pathogenesis of ERM
ILM peeling has been known to prevent post-surgical ERM growth in complicated RRD cases accompanied by proliferative vitreoretinopathy (PVR), silicone oil tamponade or diabetic retinopathy.9-11 Recently, ILM peeling has also been advocated as a procedure to prevent recurrence of ERMs after surgical removal of idiopathic ERMs.12
Authors have proposed that pathogenesis of ERMs is in association with residual vitreous cortex13 and remnant cell components,14,15 both of which are observed on the ILM surface. Histopathologically, various types of cells have been proven to exist in ERMs.15 While controversy surrounds the difference in cell distribution between idiopathic ERMs and other types of secondary ERMs or PVR membranes,14,15 authors have assumed that post-RRD ERMs comprised of retinal pigment epithelial (RPE) cells leak into the vitreous cavity
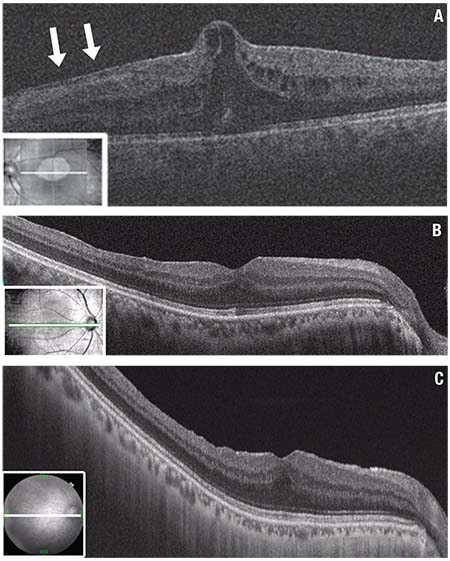 |
| Figure 1. Optical coherence tomography (OCT) shows severe epiretinal membrane (ERM) growth after vitrectomy for bilateral macula-on rhegmatogenous retinal detachment (RRD) repair in a 58-year-old man. The left eye was treated using a conventional method without internal limiting membrane (ILM) peeling. A severe epiretinal membrane developed suddenly two months later (A), which required surgical removal because visual acuity had dropped to 0.5 (20/40). Subsequently the right eye, treated with ILM peeling showed no ERM growth for more than three years and retained best-corrected visual acuity of 1.2 (≈20/16). OCT scans show the right eye at one month (B) and three years (C) postoperatively. These images present similar configurations in terms of the thickness and integrity of each layer, except for the presence of retinal dimpling at three years. (Figure 1A used with permission Elsevier: American Journal of Ophthalmology) |
residual vitreous cortex;
cell components; and
a scaffold for cellular proliferation.
Triamcinolone acetonide staining can facilitate complete removal of vitreous cortex,13 whereas the other two factors still remain unsolved. In fact, ERM growth has sometimes been confirmed after RRD repair in which complete removal of vitreous cortex has been ensured using TA.1,2 For further prevention of post-surgical ERM growth by eliminating cell components and the scaffold itself, some investigators have suggested using ILM peeling.1,2,6
How Efficacious Is ILM Peeling?
In a retrospective review of 102 consecutive cases that underwent initial vitrectomy for RRDs and were observed for at least six months postoperatively, we reported a significant preventive effect of ILM peeling on post-surgical ERM development.2 ILM was peeled with or without the assistance of TA staining (Video available at bitly.ly/RS_ILM_004). We did not use indocyanine green because of its potential to damage macular function.
Of the 102 cases in our study, 58 were treated with ILM peeling and 44 without. We confirmed ERM growth in 21 cases treated without ILM peeling, and observed no ERM growth in any cases with ILM peeling (p<0.001). Ten of the 21 cases had severe ERM growth that caused significant visual loss ≥ 0.2 logMAR (logarithm of the minimum angle of resolution) compared to the highest visual acuity obtained before detection of ERM. Nine of these cases (8.8 percent of all total cases and 20.4 percent of ILM-not-peeled cases) underwent a subsequent surgery to remove the post-surgical ERMs.
The prevalence of severe ERM growth was also significantly higher in cases without ILM peeling (p<0.001, not discussed in this article). Thus, our results strongly indicate that ILM peeling is efficacious in preventing post-RRD ERM growth, either in terms of severe ERMs (10 cases) or any ERMs (21 cases) including asymptomatic ones.
Safety of ILM Peeling
In our study, best-corrected visual acuities of cases treated with ILM peeling were not inferior to those of cases without ILM peeling, either at baseline or at the final examination.2 Other investigators have reported similar results.1,6
Additionally, we conducted further analyses of the visual prognosis after vitrectomy for macula-sparing RRDs to evaluate
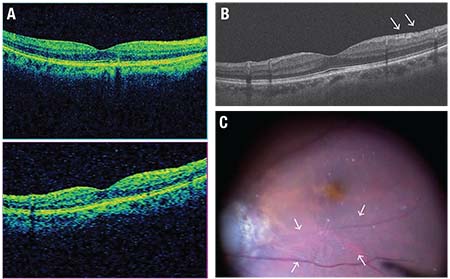 |
| Figure 2. Preoperative optical coherence tomography images and an intraoperative view of a patient with macula-sparing rhegmatogenous retinal detachment in the right eye show wrinkling of the inner surface of the macula, which looks normal on the cube protocol OCT scan (A), whereas a thin membrane with fine folds (arrows) is present on the five-line raster protocol vertical scan representing a primitive form of an epiretinal membrane (ERM) (B). The photo captured from intraoperative observation using a meniscus contact lens shows fine wrinkling on the surface of the ILM (C, surrounded by arrows). In this case, the wrinkling and the primitive ERM formation can be observed rather distinctly, but there are many cases in which the wrinkling is too fine to be captured by OCT scan or observed by a wide-viewing operating system. In order to avoid missing such findings that might later progress to ERM growth, precise observation under appropriate magnification is essential. |
Our study found no severe complications related to ILM peeling such as an iatrogenic retinal break in the macula, a macular hole (MH) or submacular hemorrhage. Although we observed in many of our cases a well-known morphologic change after ILM peeling, dissociated optic nerve fiber layer (DONFL) appearance or inner retinal dimpling (Figure 1C), it did not lead to a decline of post-surgical BCVA, which is in line with previous reports.18,19 ILM peeling is unlikely to cause severe complications or adverse impact on post-surgical vision.
ERM Growth MH Formation After RRD Repair
A macular hole is a rare but vision-threatening complication after RRD repair. Although the pathogenesis of post-surgical MH formation is still unclear, a recent report has suggested a relationship between post-RRD MH formation and ERM growth.20 In this regard, ILM peeling could prevent MH formation as well as ERM growth after vitrectomy for RRD.
ERM growth, which has been proposed to cause tangential traction at the fovea, is usually detected by its typical appearance on slit lamp examination and confirmed on optical coherence tomography (OCT) images.
However, an ERM (or thickened residual vitreous cortex) could occasionally exist where neither OCT nor a slit lamp exam reveals its presence ; it may only be identified during vitrectomy to repair secondary MH. This suggests that ERM growth may be more common after RRD repair than we thought based on observation using OCT scans. Clinically, undetectable ERMs usually remain harmless and do not require surgical intervention, whereas they could potentially play an important role in the pathogenesis of post-surgical MH formation.
Who Will Benefit From ILM Peeling?
Our data imply that ILM peeling could be considered as a prophylaxis for ERM development for any patient undergoing vitrectomy for RRD, unless the surgeon notes some unfavorable conditions. These include:
instability of the patient’s eye or head;
poor intraoperative visibility; or
extreme fragility of the retina.
However, controversies remain with regard to the technical difficulty of this procedure. When comparing common RRD to MHRD and foveal schisis associated with pathological myopia, both of which are familiar to vitreoretinal surgeons, ILM peeling in macula-involving RRD is generally easier than in MHRD. In macula-sparing cases, ILM peeling is much easier than in foveal schisis.
| Take-home Point Prevention of epiretinal membrane (ERM) growth after rhegmatogenous retinal detachment (RRD) repair is desirable because the condition may cause persistent visual impairment even after surgical removal. Internal limiting membrane (ILM) peeling prevents ERM growth effectively, with favorable visual outcomes without severe complications. Wrinkling of the ILM surface might be considered to be a sign of a primitive form of ERM growth and may be a marker that a patient would benefit from ILM peeling. |
On the other hand, one cannot completely exclude the potential adverse effect of ILM peeling on long-term visual outcomes after several decades, as several investigators have discussed in association with morphological alterations.19, 21–23 To avoid this potential risk, surgeons can apply ILM peeling to selected cases that are more likely to develop an ERM. Although some investigators have tried to identify the risk factors for ERM growth in terms of the extension of RRD, types and location of retinal tears and macular involvement, they have yet to reach a consensus.
Surface wrinkling retinopathy is a term used to describe the wrinkling of the inner retinal surface that is accompanied by ERM.24 Similar wrink-ling is sometimes detected on the macula by careful observation using a meniscus contact lens during vitrectomy for RRDs (Figure 2).
No evidence exists so far to relate this finding to subsequent ERM growth, but wrinkling could represent its earliest sign, provided that “surface wrinkling is never seen without an epiretinal membrane,” as Albert M. Roth, MD, and Robert Y. Foos, MD, reported in the 1970s before OCT was available.24 Then, ILM peeling may have been most beneficial for eyes having this phenomenon. Currently, we are peeling ILM in selected cases based on the presence of this early sign, although the number of cases is not large enough to draw a conclusion.
We should note that surface wrinkling is not always detected by OCT scans (Figure 2). Therefore, appropriate magnification and resolution is essential in order to observe the macula meticulously during vitrectomy, lest it go undetected. RS
REFERENCES
1. Nam KY, Kim JY. Effect of internal limiting membrane peeling on the development of epiretinal membrane after pars plana vitrectomy for primary rhegmatogenous retinal detachment. Retina. 2015; 35:880-885.
2. Akiyama K, Fujinami K, Watanabe K, Tsunoda K, Noda T. Internal limiting membrane peeling to prevent post-vitrectomy epiretinal membrane development in retinal detachment. Am J Ophthalmol. 2016; 171:1-10.
3. Campo RV, Sipperley JO, Sneed SR, Park DW, Dugel PU, Jacobsen J, et al. Pars plana vitrectomy without scleral buckle for pseudophakic retinal detachments. Ophthalmology. 1999; 106:1811-1815; discussion 1816.
4. Thompson JA, Snead MP, Billington BM, Barrie T, JR Thompson JR, Sparrow JM. National audit of the outcome of primary surgery for rhegmatogenous retinal detachment. II. Clinical outcomes. Eye (Lond). 2002; 16:771–777.
5. Martinez-Castillo V, Boixadera A, Distefano L, Zapata M, Garcia-Arumi J. Epiretinal membrane after pars plana vitrectomy for primary pseudophakic or aphakic rhegmatogenous retinal detachment: incidence and outcomes. Retina. 2012; 32:1350-1355.
6. Rao RC, Blinder KJ, Smith BT, Shah GK. Internal limiting membrane peeling for primary rhegmatogenous retinal detachment repair. Ophthalmology. 2013; 120:1102-1103 e1101-1102.
7. Council MD, Shah GK, Lee HC, Sharma S. Visual outcomes and complications of epiretinal membrane removal secondary to rhegmatogenous retinal detachment. Ophthalmology. 2005; 112:1218-1221.
8. Weng CY, Gregori NZ, Moysidis SN, Shi W, Smiddy WE, Flynn HW, Jr. Visual and anatomical outcomes of macular epiretinal membrane peeling after previous rhegmatogenous retinal detachment repair. Retina. 2015; 35:125-135.
9. Michalewska Z, Bednarski M, Michalewski J, Jerzy N. The role of ILM peeling in vitreous surgery for proliferative diabetic retinopathy complications. Ophthalmic Surg Lasers Imaging Retina. 2013; 44:238-242.
10. Odrobina D, Bednarski M, Cisiecki S, Michalewska Z, Kuhn F, Phd JN. Internal limiting membrane peeling as prophylaxis of macular pucker formation in eyes undergoing retinectomy for severe proliferative vitreoretinopathy. Retina. 2012; 32:226-231.
11. Aras C, Arici C, Akar S, Muftuoglu G, Yolar M, Arvas S, et al. Peeling of internal limiting membrane during vitrectomy for complicated retinal detachment prevents epimacular membrane formation. Graefes Arch Clin Exp Ophthalmol. 2009; 247:619-623.
12. Azuma K, Ueta T, Eguchi S, Aihara M. Effects of internal limiting membrane peeling combined with removal of idiopathic epiretinal membrane: a systematic review of literature and meta-analysis. Retina. 2017 Feb. 15. doi:10.1097/IAE.0000000000001537. [E pub ahead of print]
13. Sonoda KH, Sakamoto T, Enaida H, Miyazaki M, Noda Y, Nakamura T, et al. Residual vitreous cortex after surgical posterior vitreous separation visualized by intravitreous triamcinolone acetonide. Ophthalmology. 2004; 111:226-230.
14. Snead DR, James S, Snead MP. Pathological changes in the vitreoretinal junction epiretinal membrane formation. Eye (Lond). 2008; 22:1310-1317.
15. Oberstein SY, Byun J, Herrera D, Chapin EA, Fisher SK, Lewis GP. Cell proliferation in human epiretinal membranes: Characterization of cell types and correlation with disease condition and duration. Mol Vis. 2011; 17:1794-1805.
16. Clarkson JG, Green WR, Massof D. A histopathologic review of 168 cases of preretinal membrane. Am J Ophthalmol. 1977; 84:1-17.
17 Akiyama K. Effect of internal limiting membrane peeling on the post-vitrectomy epiretinal membrane growth in macula-sparing retinal detachment. Paper presented at: ARVO Asia 2017; February 8, 2017; Brisbane, Australia.
18. Tadayoni R, Paques M, Massin P, Mouki-Benani S, Mikol J, Gaudric A. Dissociated optic nerve fiber layer appearance of the fundus after idiopathic epiretinal membrane removal. Ophthalmology. 2001; 108:2279-2283.
19. Spaide RF. “Dissociated optic nerve fiber layer appearance” after internal limiting membrane removal is inner retinal dimpling. Retina. 2012; 32:1719-1726.
20. Khurana RN, Wykoff CC, Bansal AS, Akiyama K, Palmer JD, Chen E, et al. The association of epiretinal membrane with macular hole formation after rhegmatogenous retinal detachment repair. Retina. 2017; 37:1073-1078.
21. Tadayoni R, Svorenova I, Erginay A, Gaudric A, Massin P. Decreased retinal sensitivity after internal limiting membrane peeling for macular hole surgery. Br J Ophthalmol. 2012; 96:1513-1516.
22. Hisatomi T, Tachibana T, Notomi S, Koyanagi Y, Murakami Y, Takeda A, et al. Internal limiting membrane peeling-dependent retinal structural changes after vitrectomy in rhegmatogenous retinal detachment. Retina. 2017 Feb. 23. doi:10.1097/IAE.0000000000001558. [Epub ahead of print]
23. Morescalchi F, Costagliola C, Gambicorti E, Duse S, Romano MR, Semeraro F. Controversies over the role of internal limiting membrane peeling during vitrectomy in macular hole surgery. Surv Ophthalmol. 2017; 62:58-69.
24. Roth AM, Foos RY. Surface wrinkling retinopathy in eyes enucleated at autopsy. Trans Am Acad Ophthalmol Otolaryngol. 1971; 75:1047-1058.



