| ABOUT THE AUTHORS | |
 | Dr. Leng is director of ophthalmic diagnostics at the Byers Eye Institute at Stanford University, Palo Alto, Calif., and a clinical assistant professor of ophthalmology at the Stanford University School of Medicine. His e-mail is [email protected] and Twitter handle @tedleng. DISCLOSURES: Dr. Leng has no relevant disclosures. |
Determining risk factors for AMD dry-to-wet progression has implications for how frequently we follow patients and when we initiate treatment. A patient with dry AMD who presents with macular pigment change and bilateral large drusen has a 50-percent risk of progression at five years and a 67-percent risk at 10 years,2 but five- and 10-year risk scores do little to help us determine when in the next 12 months we should be seeing this patient again.
With such high long-term progression risks, should we see these patients again in 12, six or three months? Or, should we see them monthly?
Features That Comprise AMD Prediction Model |
| Demographic Features: • Age • Sex • Status of fellow eye (dry or wet) Imaging Features: • Number of separate drusen regions • Mean volume per drusen region • Total volume of drusen regions • Mean area per drusen region • Total area of drusen regions • Drusen density in affected area • Extent of drusen-affected area • Maximum drusen height • Mean drusen slope • Mean reflectivity inside drusen regions • Standard deviation of reflectivity inside drusen regions |
Traditional Techniques
While seeing all intermediate dry AMD patients every three months for a dilated fundus examination and spectral-domain optical coherence tomography (SD-OCT) imaging would likely allow for early detection of all wet AMD conversions and minimal delay to a first anti-VEGF injection, this strategy is not without its shortcomings. It is costly, both in terms of dollars spent on evaluation and management examinations and imaging CPT codes, as well as in time patients spend in doctors’ offices.Also, our clinics can only handle a maximum number of patients, and seeing all intermediate dry AMD patients every three months would reduce the available slots for non-AMD retina patients with active disease. Further, it is not efficient to see every patient whose eyes may be at high risk for progression because we would be examining a lot of stable patients without any disease to treat.
Adding genetic testing to the clinical examination is one way to boost the power of prediction. The presence of certain high-risk, single-nucleotide polymorphisms (SNPs) may indicate eyes at risk of progression and, therefore, patients who require more frequent monitoring.
Using Big Data
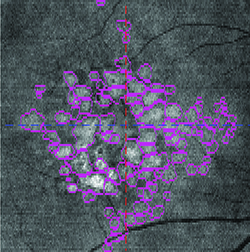 | 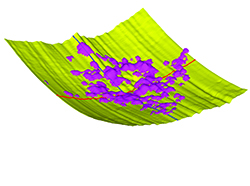 |
| Figure 1. Outputs from a drusen segmentation algorithm designed by our group. Two-dimensional (left) and three-dimensional (above) maps of drusen in an eye with intermediate dry age-related macular degeneration (AMD). These segmentations were used to measure 11 different imaging biomarkers that may be predictive of AMD progression. |
Each SD-OCT scan has more data in the cube than can be manually analyzed during the time of an average clinical encounter. Specifically, a typical SD-OCT scan contains more than 67 million points of data (to construct a cube scan, the typical OCT platform performs 512 A-scans per B-scan, with each individual A-scan collecting 1,024 data points; 128 B-scans make the typical cube). Unfortunately, we use less than 1 percent of the data from these scans during the visit.
Ideally, we could analyze all of the data in each OCT scan in an automated fashion and use the output to make predictions about current and future disease activity. Moreover, the method for doing this type of automatic analysis should be device agnostic so that we could input OCT data from any manufacturer’s device to get the analysis. Finally, the method for OCT analysis should be able to process OCT scans of poor quality and analyze scans that have intermediate to advanced macular disease.
Interestingly, the analytical software packages that come with most commercial OCT devices do not take full advantage of all the data each OCT scan contains. The interpretation algorithms most platforms use measure a few parameters but cannot perform in-depth measurements of multiple retinal imaging features. Thus, if a retina specialist wants to measure more than three or four biomarkers in an OCT scan, then he or she would have to create an algorithm using the raw imaging data.
Designing New Tools
 |
| Figure 2. Horizontal spectral-domain optical coherence tomography B-scans from two eyes with high-risk characteristics for age-related macular degeneration (AMD) progression. Both scans demonstrate increased drusen height, slope and reflectivity—all features found in our study to increase the risk for AMD progression. Both of these eyes eventually progressed to wet AMD. |
Typically, drusen must be identified after images are cleaned up. Because we were not satisfied with the commercially available software for identifying drusen, we created an automated segmentation algorithm to extract drusen features from de-noised images (Figure 1, page 21).5 We also designed the algorithms to work with images from any vendor and have successfully segmented images from devices made by Carl Zeiss Meditec, Heidelberg and Bioptigen.
Imaging Biomarkers
That a larger total drusen area and volume in the macula would increase the risk for AMD progression has been a long-held assumption, but those are only two possible drusen biomarkers that one could consider in a statistical model to predict which eyes with AMD were at risk for progression. Our research team created a model that incorporates 11 separate drusen biomarkers identified on imaging in an AMD prediction model.6 When combined with the change in those biomarkers over time as well as with demographic information, our predictive model assesses 26 total features (Table, page 21).We tested our model on a retrospective dataset from our patient population at the Byers Eye Institute at Stanford University. In all, the dataset included 330 eyes from 244 patients over a five-year period. A total of 2,146 SD-OCT scans were de-noised, segmented and had imaging biomarkers extracted.
We knew that 36 eyes in this dataset progressed to wet AMD during the study period, so we compared the 26 features in a statistical model between the eyes that progressed to those that remained stable. We found that the area, volume, height and reflectivity features of drusen were most important in distinguishing between progressing and nonprogressing cases (Figure 2). The patient’s age and dry/wet status of the fellow eye were also significant. After the analysis identified the most relevant features, a predictive model for AMD progression was created.
One unique characteristic of this model is that it could be used to calculate progression risk for any timespan. Unlike the Age-Related Eye Disease Study (AREDS) and genetic risk models that determine risk over multi-year time spans, our model could predict the risk of progression within three, six or 12 months—which is what retina specialists and patients are probably most interested in knowing.
If a retina specialist knew there was a high risk of an intermediate dry AMD eye progressing in six months, he or she would likely monitor that patient more closely than one that had a low risk of progressing over six months. With our algorithm, we hope to bring this sort of real-time decision support to the clinic.
The Future
The Role of Big Data in Home Monitoring |
| Home monitoring can also play a role in collecting data to track disease progression. The HOME Study—for Home Monitoring of Eye—is a randomized trial that compared patients’ home use of the prescription-based ForeseeHome (Notal Vision Inc.) device plus standard care vs. standard care alone to monitor for choroidal neovascularization in wet AMD.7 Among 1,520 subjects, participants in the device arm demonstrated a smaller decline in visual acuity with fewer letters lost from baseline to detection of choroidal neovascularization, a median loss of 4 letters vs. 9 letters, respectively. ForeseeHome transmits test results to a central monitoring center to alert physicians of any significant visual changes. The smartphone-based Sightbook (DigiSight Technologies) is another remote application that shares results of a variety of patient-administered vision tests in real time with physician’s office and sends reminders to patients.8 This application was found to have a high level of agreement with Early Treatment for Diabetic Retinopathy Study (ETDRS) distance visual acuity and near vision testing.9 My VisionTrack (Vital Art and Science Inc.) is a another prescription-only smartphone app that uses a shape discrimination algorithm that stores test results in a cloud database for physician and patient access. Again, a study has found these test results to correlate well with standard VA tests.10 |
To address these issues, we are in the process of using other retrospective AMD OCT datasets to validate the predictive features of our model. We are also beginning to incorporate genetic risk factors into our statistical model, which we expect will boost our predictive accuracy.
Additionally, we are enrolling subjects into a prospective clinical trial (NCT02422160) at the Byers Eye Institute that will follow eyes with dry AMD longitudinally with regular SD-OCT scans, incorporate demographic and genetic data and provide real-time risk assessment for patient management. Each time an eye gets an OCT scan, the algorithm will extract imaging biomarker features and integrate them into the risk assessment model for that patient, giving progression risk information that the clinician can use at the point of care to decide which patients to follow closely. This is the last step in solidifying this type of clinical decision support system. RS
References
1. Arnold JJ, Campain A, Barthelmes D, et al. Two-year outcomes of “treat and extend” intravitreal therapy for neovascular age-related macular degeneration. Ophthalmology. 2015; 122:1212-1219.
2. Age-Related Eye Disease Study Research Group. A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins c, and e, beta carotene, and zinc for age-related macular degeneration and vision loss: AREDS Report No. 8. Arch Ophthalmol. 2001;119:1417-1436.
3. Eldaly MA, Styles C. First versus second eye intravitreal ranibizumab therapy for wet AMD. Retina. 2009;29:325-328.
4. Chen Q, de Sisternes L, Leng T, Rubin DL. Application of improved homogeneity similarity based denoising in optical coherence tomography retinal images. J Digital Imaging. 2015;28:346-361.
5. Chen Q, Leng T, Zheng L, et al. Automated drusen segmentation and quantification in SD-OCT images. Med Image Anal. 2013;17:1058-1072.
6. De Sisternes L, Simon N, Tibshirani R, et al. Quantitative SD-OCT imaging biomarkers as indicators of age-related macular degeneration progression. Invest Ophthalmol Vis Sci. 2014;55:7093-7109.
7. AREDS2-HOME Study Research Group, Chew EY, Clemons TE, Bressler SB, et al. Randomized trial of a home monitoring system for early detection of choroidal neovascularization home monitoring of the Eye (HOME) study. Ophthalmology. 2014 Feb;121(2):535-544.
8. Fung A. Smartphone based home monitoring. Presented at: American Academy of Ophthalmology 2014 Retina Subspecialty Day; October 18, 2014; Chicago, IL.
9. Yu S-Y, et al. Reliability of smartphone-based electronic visual acuity testing. Poster presented at the ARVO 2014 Annual Meeting; May 2014; Orlando, FL.
10. Wang YZ, He YG, Mitzel G, Zhang S, Bartlett M. Handheld shape discrimination hyperacuity test on a mobile device for remote monitoring of visual function in maculopathy. Invest Ophthalmol Vis Sci. 2013; 54:5497-5504.
2. Age-Related Eye Disease Study Research Group. A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins c, and e, beta carotene, and zinc for age-related macular degeneration and vision loss: AREDS Report No. 8. Arch Ophthalmol. 2001;119:1417-1436.
3. Eldaly MA, Styles C. First versus second eye intravitreal ranibizumab therapy for wet AMD. Retina. 2009;29:325-328.
4. Chen Q, de Sisternes L, Leng T, Rubin DL. Application of improved homogeneity similarity based denoising in optical coherence tomography retinal images. J Digital Imaging. 2015;28:346-361.
5. Chen Q, Leng T, Zheng L, et al. Automated drusen segmentation and quantification in SD-OCT images. Med Image Anal. 2013;17:1058-1072.
6. De Sisternes L, Simon N, Tibshirani R, et al. Quantitative SD-OCT imaging biomarkers as indicators of age-related macular degeneration progression. Invest Ophthalmol Vis Sci. 2014;55:7093-7109.
7. AREDS2-HOME Study Research Group, Chew EY, Clemons TE, Bressler SB, et al. Randomized trial of a home monitoring system for early detection of choroidal neovascularization home monitoring of the Eye (HOME) study. Ophthalmology. 2014 Feb;121(2):535-544.
8. Fung A. Smartphone based home monitoring. Presented at: American Academy of Ophthalmology 2014 Retina Subspecialty Day; October 18, 2014; Chicago, IL.
9. Yu S-Y, et al. Reliability of smartphone-based electronic visual acuity testing. Poster presented at the ARVO 2014 Annual Meeting; May 2014; Orlando, FL.
10. Wang YZ, He YG, Mitzel G, Zhang S, Bartlett M. Handheld shape discrimination hyperacuity test on a mobile device for remote monitoring of visual function in maculopathy. Invest Ophthalmol Vis Sci. 2013; 54:5497-5504.



